FoxO3a regulates erythroid differentiation and induces BTG1, an activator of protein arginine methyl transferase 1
- PMID: 14734530
- PMCID: PMC2172323
- DOI: 10.1083/jcb.200307056
FoxO3a regulates erythroid differentiation and induces BTG1, an activator of protein arginine methyl transferase 1
Abstract
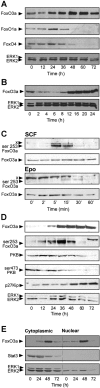
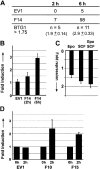
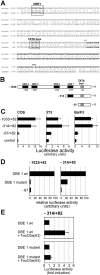
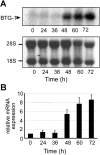
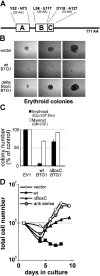
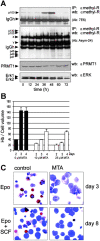
Erythropoiesis requires tight control of expansion, maturation, and survival of erythroid progenitors. Because activation of phosphatidylinositol-3-kinase (PI3K) is required for erythropoietin/stem cell factor-induced expansion of erythroid progenitors, we examined the role of the PI3K-controlled Forkhead box, class O (FoxO) subfamily of Forkhead transcription factors. FoxO3a expression and nuclear accumulation increased during erythroid differentiation, whereas untimely induction of FoxO3a activity accelerated differentiation of erythroid progenitors to erythrocytes. We identified B cell translocation gene 1 (BTG1)/antiproliferative protein 2 as a FoxO3a target gene in erythroid progenitors. Promoter studies indicated BTG1 as a direct target of FoxO3a. Expression of BTG1 in primary mouse bone marrow cells blocked the outgrowth of erythroid colonies, which required a domain of BTG1 that binds protein arginine methyl transferase 1. During erythroid differentiation, increased arginine methylation coincided with BTG1 expression. Concordantly, inhibition of methyl transferase activity blocked erythroid maturation without affecting expansion of progenitor cells. We propose FoxO3a-controlled expression of BTG1 and subsequent regulation of protein arginine methyl transferase activity as a novel mechanism controlling erythroid expansion and differentiation.
Figures

References
-
- Bao, H., S.M. Jacobs-Helber, A.E. Lawson, K. Penta, A. Wickrema, and S.T. Sawyer. 1999. Protein kinase B (c-Akt), phosphatidylinositol 3-kinase, and STAT5 are activated by erythropoietin (EPO) in HCD57 erythroid cells but are constitutively active in an EPO-independent, apoptosis-resistant subclone (HCD57-SREI cells). Blood. 93:3757–3773. - PubMed
-
- Berthet, C., F. Guehenneux, V. Revol, C. Samarut, A. Lukaszewicz, C. Dehay, C. Dumontet, J.P. Magaud, and J.P. Rouault. 2002. Interaction of PRMT1 with BTG/TOB proteins in cell signalling: molecular analysis and functional aspects. Genes Cells. 7:29–39. - PubMed
-
- Beug, H., S. Palmieri, C. Freudenstein, H. Zentgraf, and T. Graf. 1982. Hormone-dependent terminal differentiation in vitro of chicken erythroleukemia cells transformed by ts mutants of avian erythroblastosis virus. Cell. 28:907–919. - PubMed
-
- Bogdan, J.A., C. Adams-Burton, D.L. Pedicord, D.A. Sukovich, P.A. Benfield, M.H. Corjay, J.K. Stoltenborg, and I.B. Dicker. 1998. Human carbon catabolite repressor protein (CCR4)-associative factor 1: cloning, expression and characterization of its interaction with the B-cell translocation protein BTG1. Biochem. J. 336:471–481. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

