Cdc6 chromatin affinity is unaffected by serine-54 phosphorylation, S-phase progression, and overexpression of cyclin A
- PMID: 14749377
- PMCID: PMC344196
- DOI: 10.1128/MCB.24.4.1614-1627.2004
Cdc6 chromatin affinity is unaffected by serine-54 phosphorylation, S-phase progression, and overexpression of cyclin A
Abstract
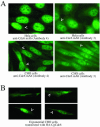
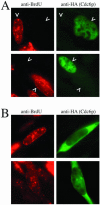
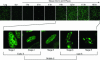
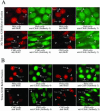
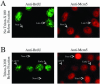
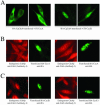
Ectopically expressed Cdc6 is translocated from the nucleus during S phase in a cyclin A-Cdk2-dependent process, suggesting that reinitiation of DNA replication is prevented by removal of phosphorylated Cdc6 from chromatin after origin firing. However, whether endogenous Cdc6 translocates during S phase remains controversial. To resolve the questions regarding regulation of endogenous Cdc6, we cloned the cDNA encoding the Chinese hamster Cdc6 homolog and specifically focused on analyzing the localizations and chromatin affinities of endogenous and exogenous proteins during S phase and following overexpression of cyclin A. In agreement with other reports, ectopically expressed Cdc6 translocates from the nucleus during S phase and in response to overexpressed cyclin A. In contrast, using a combination of biochemical and immunohistochemical assays, we show convincingly that endogenous Cdc6 remains nuclear and chromatin bound throughout the entire S period, while Mcm5 loses chromatin affinity during S phase. Overexpression of cyclin A is unable to alter the nuclear localization of Cdc6. Furthermore, using a phosphospecific antibody we show that phosphoserine-54 Cdc6 maintains a high affinity for chromatin during the S period. Considering recent in vitro studies, these data are consistent with a proposed model in which Cdc6 is serine-54 phosphorylated during S phase and functions as a chromatin-bound signal that prevents reformation of prereplication complexes.
Figures



References
-
- Alcasabas, A. A., A. J. Osborn, J. Bachant, F. Hu, P. J. Werler, K. Bousset, K. Furuya, J. F. Diffley, A. M. Carr, and S. J. Elledge. 2001. Mrc1 transduces signals of DNA replication stress to activate Rad53. Nat. Cell Biol. 3:958-965. - PubMed
-
- Bell, S. P., and A. Dutta. 2002. DNA replication in eukaryotic cells. Annu. Rev. Biochem. 71:333-374. - PubMed
-
- Bell, S. P., and B. Stillman. 1992. ATP-dependent recognition of eukaryotic origins of DNA replication by a multiprotein complex. Nature 357:128-134. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
