Arachidonoyl-phospholipid remodeling in proliferating murine T cells
- PMID: 14754461
- PMCID: PMC343295
- DOI: 10.1186/1476-511X-3-1
Arachidonoyl-phospholipid remodeling in proliferating murine T cells
Abstract
Background: Previous studies have shown that the functional capacity of T cells may be modulated by the composition of fatty acids within, and the release of fatty acids from membrane phospholipids, particularly containing arachidonic acid (AA). The remodeling of AA within membrane phospholipids of resting and proliferating CD4+ and CD8+ T cells is examined in this study.
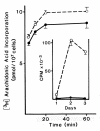
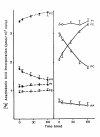
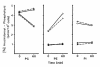
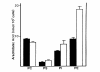
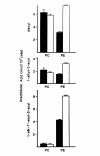
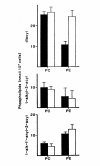
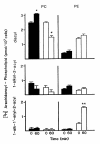
Results: Splenic T cells were cultured in the presence or absence of anti-CD3 mAb for 48 h then labeled with [3H]AA for 20 min. In unstimulated cells, labeled AA was preferentially incorporated into the phosphoglycerides, phosphatidylcholine (PC) followed by phosphatidylinositol (PI) and phosphatidylethanolamine (PE). During a subsequent chase in unlabeled medium unstimulated CD4+ and CD8+ T cells demonstrated a significant and highly selective transfer of free, labeled AA into the PC pool. In contrast, proliferating CD4+ and CD8+ T cells distributed labeled [3H]AA predominantly into PI followed by PC and PE. Following a chase in AA-free medium, a decline in the content of [3H]AA-PC was observed in association with a comparable increase in [3H]AA-PE. Subsequent studies revealed that the cold AA content of all PE species was increased in proliferating T cells compared with that in non-cycling cells, but that enrichment in AA was observed only in the ether lipid fractions. Finally, proliferating T cells preincubated with [3H]AA exhibited a significant loss of labeled arachidonate in the PC fraction and an equivalent gain in labeled AA in 1-alk-1'-enyl-2-arachidonoyl-PE during a chase in unlabeled medium.
Conclusion: This apparent unidirectional transfer of AA from PC to ether-containing PE suggests the existence of a CoA-independent transacylase system in T cells and supports the hypothesis that arachidonoyl phospholipid remodeling may play a role in the regulation of cellular proliferation.
Figures
Similar articles
-
Rapid remodeling of arachidonate from phosphatidylcholine to phosphatidylethanolamine pools during mast cell activation.J Immunol. 1992 Mar 15;148(6):1784-91. J Immunol. 1992. PMID: 1541818
-
Remodeling of arachidonate-containing phosphoglycerides within the human neutrophil.J Biol Chem. 1986 Jun 15;261(17):7771-7. J Biol Chem. 1986. PMID: 3086318
-
Comparison of acceptor and donor substrates in the CoA-independent transacylase reaction in human neutrophils.Biochim Biophys Acta. 1996 May 20;1300(3):187-96. doi: 10.1016/0005-2760(96)00011-2. Biochim Biophys Acta. 1996. PMID: 8679683
-
Relationship between arachidonate--phospholipid remodeling and apoptosis.Biochemistry. 1996 Jul 16;35(28):9187-96. doi: 10.1021/bi9530245. Biochemistry. 1996. PMID: 8703924
-
Role of Ca2+ in phosphatidylinositol response and arachidonic acid release in formylated tripeptide- or Ca2+ ionophore A23187-stimulated guinea pig neutrophils.J Immunol. 1983 Jun;130(6):2849-55. J Immunol. 1983. PMID: 6406597
Cited by
-
Phospholipid remodeling and eicosanoid signaling in colon cancer cells.Indian J Biochem Biophys. 2014 Dec;51(6):512-9. Indian J Biochem Biophys. 2014. PMID: 25823224 Free PMC article. Review.
-
Molecular dynamics simulations of unsaturated lipid bilayers: effects of varying the numbers of double bonds.Eur Biophys J. 2005 Jun;34(4):294-305. doi: 10.1007/s00249-004-0455-7. Epub 2005 Feb 2. Eur Biophys J. 2005. PMID: 15688184
-
Immunotoxicity assessment of rice-derived recombinant human serum albumin using human peripheral blood mononuclear cells.PLoS One. 2014 Aug 6;9(8):e104426. doi: 10.1371/journal.pone.0104426. eCollection 2014. PLoS One. 2014. PMID: 25099245 Free PMC article.
-
Fatty acid remodeling in cellular glycerophospholipids following the activation of human T cells.J Lipid Res. 2013 Oct;54(10):2665-77. doi: 10.1194/jlr.M037044. Epub 2013 Jul 26. J Lipid Res. 2013. PMID: 23894206 Free PMC article.
-
Epigenetics: The New Frontier in the Landscape of Asthma.Scientifica (Cairo). 2016;2016:4638949. doi: 10.1155/2016/4638949. Epub 2016 May 11. Scientifica (Cairo). 2016. PMID: 27293973 Free PMC article. Review.
References
-
- Gill R, Clark W. Membrane structure-function relationships in cell-mediated cytolysis. I. Effect of exogenously incorporated fatty acids on effector cell function in cell-mediated cytolysis. J Immunol. 1980;125:689–695. - PubMed
-
- Goetzl EJ. Mediators of immediate hypersensitivity derived from arachidonic acid. N Engl J Med. 1980;303:822–825. - PubMed
-
- Sekiguchi K, Tsukuda M, Ogita K, Kikkawa U, Nishizuka Y. Three distinct forms of rat brain protein kinase C: differential response to unsaturated fatty acids. Biochem Biophys Res Commun. 1987;145:797–802. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

