Inositol-1,4,5-trisphosphate-dependent Ca(2+) signalling in cat atrial excitation-contraction coupling and arrhythmias
- PMID: 14754996
- PMCID: PMC1664857
- DOI: 10.1113/jphysiol.2003.058529
Inositol-1,4,5-trisphosphate-dependent Ca(2+) signalling in cat atrial excitation-contraction coupling and arrhythmias
Abstract
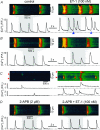
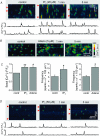
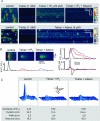
Inositol-1,4,5-trisphosphate (IP(3))-dependent Ca(2+) release represents the major Ca(2+) mobilizing pathway responsible for diverse functions in non-excitable cells. In the heart, however, its role is largely unknown or controversial. In intact cat atrial myocytes, endothelin (ET-1) increased basal [Ca(2+)](i) levels, enhanced action potential-evoked [Ca(2+)](i) transients, caused [Ca(2+)](i) transients with alternating amplitudes (Ca(2+) alternans), and facilitated spontaneous Ca(2+) release from the sarcoplasmic reticulum (SR) in the form of Ca(2+) sparks and arrhythmogenic Ca(2+) waves. These effects were prevented by the IP(3) receptor (IP(3)R) blocker aminoethoxydiphenyl borate (2-APB), suggesting the involvement of IP(3)-dependent SR Ca(2+) release. In saponin-permeabilized myocytes IP(3) and the more potent IP(3)R agonist adenophostin increased basal [Ca(2+)](i) and the frequency of spontaneous Ca(2+) sparks. In the presence of tetracaine to eliminate Ca(2+) release from ryanodine receptor (RyR) SR Ca(2+) release channels, IP(3) and adenophostin triggered unique elementary, non-propagating IP(3)R-dependent Ca(2+) release events with amplitudes and kinetics that were distinctly different from classical RyR-dependent Ca(2+) sparks. The effects of IP(3) and adenophostin were prevented by heparin and 2-APB. The data suggest that IP(3)-dependent Ca(2+) release increases [Ca(2+)](i) in the vicinity of RyRs and thus facilitates Ca(2+)-induced Ca(2+) release during excitation-contraction coupling. It is concluded that in the adult mammalian atrium IP(3)-dependent Ca(2+) release enhances atrial Ca(2+) signalling and exerts a positive inotropic effect. In addition, by facilitating Ca(2+) release, IP(3) may also be an important component in the development of Ca(2+)-mediated atrial arrhythmias.
Figures

Similar articles
-
Inositol-1,4,5-trisphosphate induced Ca2+ release and excitation-contraction coupling in atrial myocytes from normal and failing hearts.J Physiol. 2015 Mar 15;593(6):1459-77. doi: 10.1113/jphysiol.2014.283226. Epub 2014 Dec 22. J Physiol. 2015. PMID: 25416623 Free PMC article.
-
The role of inositol 1,4,5-trisphosphate receptors in Ca(2+) signalling and the generation of arrhythmias in rat atrial myocytes.J Physiol. 2002 Jun 1;541(Pt 2):395-409. doi: 10.1113/jphysiol.2001.013411. J Physiol. 2002. PMID: 12042347 Free PMC article.
-
Endothelin-1-induced arrhythmogenic Ca2+ signaling is abolished in atrial myocytes of inositol-1,4,5-trisphosphate(IP3)-receptor type 2-deficient mice.Circ Res. 2005 Jun 24;96(12):1274-81. doi: 10.1161/01.RES.0000172556.05576.4c. Epub 2005 Jun 2. Circ Res. 2005. PMID: 15933266
-
Atrial local Ca2+ signaling and inositol 1,4,5-trisphosphate receptors.Prog Biophys Mol Biol. 2010 Sep;103(1):59-70. doi: 10.1016/j.pbiomolbio.2010.02.002. Epub 2010 Mar 1. Prog Biophys Mol Biol. 2010. PMID: 20193706 Review.
-
Local calcium gradients during excitation-contraction coupling and alternans in atrial myocytes.J Physiol. 2003 Jan 1;546(Pt 1):19-31. doi: 10.1113/jphysiol.2002.025239. J Physiol. 2003. PMID: 12509476 Free PMC article. Review.
Cited by
-
Role of inositol 1,4,5-trisphosphate in the regulation of ventricular Ca(2+) signaling in intact mouse heart.J Mol Cell Cardiol. 2012 Dec;53(6):768-79. doi: 10.1016/j.yjmcc.2012.08.019. Epub 2012 Aug 31. J Mol Cell Cardiol. 2012. PMID: 22960455 Free PMC article.
-
Paclitaxel induces calcium oscillations via an inositol 1,4,5-trisphosphate receptor and neuronal calcium sensor 1-dependent mechanism.Proc Natl Acad Sci U S A. 2006 Nov 28;103(48):18356-61. doi: 10.1073/pnas.0607240103. Epub 2006 Nov 17. Proc Natl Acad Sci U S A. 2006. PMID: 17114292 Free PMC article.
-
Calpain-dependent cleavage of junctophilin-2 and T-tubule remodeling in a mouse model of reversible heart failure.J Am Heart Assoc. 2014 Jun 23;3(3):e000527. doi: 10.1161/JAHA.113.000527. J Am Heart Assoc. 2014. PMID: 24958777 Free PMC article.
-
The type 2 inositol 1,4,5-trisphosphate receptor, emerging functions for an intriguing Ca²⁺-release channel.Biochim Biophys Acta. 2015 Sep;1853(9):1992-2005. doi: 10.1016/j.bbamcr.2014.12.006. Epub 2014 Dec 10. Biochim Biophys Acta. 2015. PMID: 25499268 Free PMC article. Review.
-
Intestinal enteroendocrine cells rely on ryanodine and IP3 calcium store receptors for mechanotransduction.J Physiol. 2023 Jan;601(2):287-305. doi: 10.1113/JP283383. Epub 2022 Dec 13. J Physiol. 2023. PMID: 36428286 Free PMC article.
References
-
- Bers DM. Excitation-Contraction Coupling and Cardiac Contractile Force. 2nd edn. Dordrecht, Netherlands: Kluwer; 2001.
-
- Bootman MD. Hormone-evoked subcellular Ca2+ signals in HeLa cells. Cell Calcium. 1996;20:97–104. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

