The role of Shc and insulin-like growth factor 1 receptor in mediating the translocation of estrogen receptor alpha to the plasma membrane
- PMID: 14764897
- PMCID: PMC357054
- DOI: 10.1073/pnas.0308334100
The role of Shc and insulin-like growth factor 1 receptor in mediating the translocation of estrogen receptor alpha to the plasma membrane
Abstract
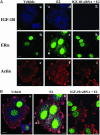
Our previous studies demonstrated that 17beta-estradiol (E2) rapidly induces the interaction of estrogen receptor alpha (ERalpha) with the adapter protein Shc, the translocation of ERalpha to the cell membrane, and the formation of dynamic membrane structures in MCF-7 breast cancer cells. The present study examined how E2 causes ERalpha to translocate to the region of the plasma membrane and focused on mechanisms whereby Shc and the insulin-like growth factor-1 receptor (IGF-1R) mediate this process. Shc physically interacts with IGF-1R in the plasma membrane, and E2 activates IGF-1R. We reasoned that ERalpha, when bound to Shc, would be directed to the region of the plasma membrane by the same processes, causing membrane translocation of Shc. We confirmed that E2 rapidly induced IGF-1R phosphorylation and demonstrated that E2 induced formation of a ternary protein complex among Shc, ERalpha, and IGF-1R. Knock down of Shc with a specific small inhibitory RNA decreased the association of ERalpha with IGF-1R by 87%, suggesting that Shc is a crucial molecule in the formation of this ternary complex. Confocal microscopy studies provided further confirmation of the functional roles of Shc and the IGF-1R in the translocation of ERalpha to the region of the membrane. Down-regulation of Shc, ERalpha, or IGF-1R with specific small inhibitory RNAs all blocked E2-induced mitogen-activated protein kinase phosphorylation. Together, our results demonstrate that Shc and IGF-1R serve as key elements in the translocation of ERalpha to the cell membrane and in the facilitation of ERalpha-mediated rapid E2 action.
Figures




References
-
- Yager, J. D. (2000) J. Natl. Cancer Inst. Monogr. 27, 67-73. - PubMed
-
- Pietras, R. J., Nemere, I. & Szego, C. M. (2001) Endocrine 14, 417-427. - PubMed
-
- Ho, K. J. & Liao, J. K. (2002) Arterioscler. Thromb. Vasc. Biol. 22, 1952-1961. - PubMed
-
- Collins, P. & Webb, C. (1999) Nat. Med. 5, 1130-1131. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

