Postsynaptic density assembly is fundamentally different from presynaptic active zone assembly
- PMID: 14960624
- PMCID: PMC6730341
- DOI: 10.1523/JNEUROSCI.3819-03.2004
Postsynaptic density assembly is fundamentally different from presynaptic active zone assembly
Abstract
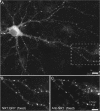
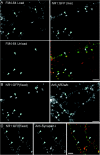
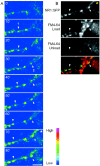
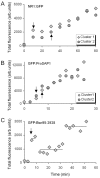
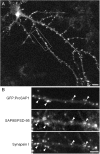
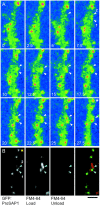
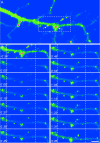
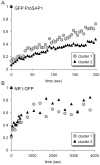
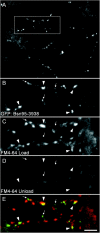
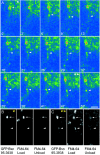
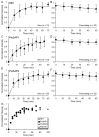
The cellular mechanisms involved in the formation of the glutamatergic postsynaptic density (PSD) are mainly unknown. Previous studies have indicated that PSD assembly may occur in situ by a gradual recruitment of postsynaptic molecules, whereas others have suggested that the PSD may be assembled from modular transport packets assembled elsewhere. Here we used cultured hippocampal neurons and live cell imaging to examine the process by which PSD molecules from different layers of the PSD are recruited to nascent postsynaptic sites. GFP-tagged NR1, the essential subunit of the NMDA receptor, and ProSAP1/Shank2 and ProSAP2/Shank3, scaffolding molecules thought to reside at deeper layers of the PSD, were recruited to new synaptic sites in gradual manner, with no obvious involvement of discernible discrete transport particles. The recruitment kinetics of these three PSD molecules were remarkably similar, which may indicate that PSD assembly rate is governed by a common upstream rate-limiting process. In contrast, the presynaptic active zone (AZ) molecule Bassoon was observed to be recruited to new presynaptic sites by means of a small number of mobile packets, in full agreement with previous studies. These findings indicate that the assembly processes of PSDs and AZs may be fundamentally different.
Figures
References
-
- Ahmari SE, Buchanan J, Smith SJ (2000) Assembly of presynaptic active zones from cytoplasmic transport packets. Nat Neurosci 3: 445-451. - PubMed
-
- Barry MF, Ziff EB (2002) Receptor trafficking and the plasticity of excitatory synapses. Curr Opin Neurobiol 12: 279-286. - PubMed
-
- Böckers TM, Mameza MG, Kreutz MR, Bockmann J, Weise C, Buck F, Richter D, Gundelfinger ED, Kreienkamp HJ (2001) Synaptic scaffolding proteins in rat brain. Ankyrin repeats of the multidomain Shank protein family interact with the cytoskeletal protein α-fodrin. J Biol Chem 276: 40104-40112. - PubMed
-
- Bockmann J, Kreutz MR, Gundelfinger ED, Boeckers TM (2002) ProSAP/Shank postsynaptic density proteins interact with insulin receptor tyrosine kinase substrate IRSp53. J Neurochem 83: 1013-1017. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
