Vaccinia virus A36R membrane protein provides a direct link between intracellular enveloped virions and the microtubule motor kinesin
- PMID: 14963148
- PMCID: PMC369226
- DOI: 10.1128/jvi.78.5.2486-2493.2004
Vaccinia virus A36R membrane protein provides a direct link between intracellular enveloped virions and the microtubule motor kinesin
Abstract
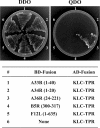
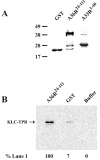
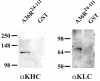
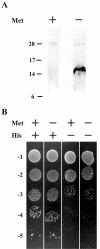
Previous work demonstrated that intracellular enveloped vaccinia virus virions use microtubules to move from the site of membrane wrapping to the cell periphery. The mechanism and direction of intracellular virion movement predicted that viral proteins directly or indirectly interact with the microtubule motor protein kinesin. The yeast two-hybrid assay was used to test for interactions between the light chain of kinesin and the cytoplasmic tails from five viral envelope proteins. We found that the N-terminal tetratricopeptide repeat region of the kinesin light chain (KLC-TPR) interacted with the cytoplasmic tail of the viral A36R protein. A series of C- and N-terminal truncations of A36R further defined a region from residues 81 to 111 that was sufficient for interaction with KLC-TPR. Interactions were confirmed by using pull-down assays with purified glutathione S-transferase (GST)-A36R and (35)S-labeled KLC-TPR. The defined region on A36R for interaction with kinesin overlaps the recently defined region (residues 91 to 111) for interaction with the A33R envelope protein. The yeast three-hybrid system was used to demonstrate that expression of A33R interrupted the interaction between A36R and KLC-TPR, indicating that the binding of A36R is mutually exclusive to either A33R or kinesin. Pull-down assays with purified GST-A36R and (35)S-labeled KLC-TPR in the presence of competing A33R corroborated these findings. Collectively, these results demonstrated that the viral A36R protein interacts directly with the microtubule motor protein kinesin and that the viral protein A33R may regulate this interaction.
Figures

Similar articles
-
Mapping and functional analysis of interaction sites within the cytoplasmic domains of the vaccinia virus A33R and A36R envelope proteins.J Virol. 2003 Apr;77(7):4113-26. doi: 10.1128/jvi.77.7.4113-4126.2003. J Virol. 2003. PMID: 12634370 Free PMC article.
-
The vaccinia virus A33R protein provides a chaperone function for viral membrane localization and tyrosine phosphorylation of the A36R protein.J Virol. 2001 Jan;75(1):303-10. doi: 10.1128/JVI.75.1.303-310.2001. J Virol. 2001. PMID: 11119600 Free PMC article.
-
Kinesin-dependent movement on microtubules precedes actin-based motility of vaccinia virus.Nat Cell Biol. 2001 Nov;3(11):992-1000. doi: 10.1038/ncb1101-992. Nat Cell Biol. 2001. PMID: 11715020
-
The formation and function of extracellular enveloped vaccinia virus.J Gen Virol. 2002 Dec;83(Pt 12):2915-2931. doi: 10.1099/0022-1317-83-12-2915. J Gen Virol. 2002. PMID: 12466468 Review.
-
Directionality of kinesin motors.Acta Biochim Pol. 2002;49(4):813-21. Acta Biochim Pol. 2002. PMID: 12545188 Review.
Cited by
-
Vaccinia virus H2 protein is an essential component of a complex involved in virus entry and cell-cell fusion.J Virol. 2005 Apr;79(8):4744-54. doi: 10.1128/JVI.79.8.4744-4754.2005. J Virol. 2005. PMID: 15795260 Free PMC article.
-
Lipid membranes in poxvirus replication.Viruses. 2010 Apr;2(4):972-986. doi: 10.3390/v2040972. Epub 2010 Apr 6. Viruses. 2010. PMID: 21994664 Free PMC article.
-
Coupling viruses to dynein and kinesin-1.EMBO J. 2011 Aug 31;30(17):3527-39. doi: 10.1038/emboj.2011.283. EMBO J. 2011. PMID: 21878994 Free PMC article. Review.
-
Effect of Clinically Used Microtubule Targeting Drugs on Viral Infection and Transport Function.Int J Mol Sci. 2022 Mar 22;23(7):3448. doi: 10.3390/ijms23073448. Int J Mol Sci. 2022. PMID: 35408808 Free PMC article.
-
Interaction between vaccinia virus extracellular virus envelope A33 and B5 glycoproteins.J Virol. 2006 Sep;80(17):8763-77. doi: 10.1128/JVI.00598-06. J Virol. 2006. PMID: 16912323 Free PMC article.
References
-
- Appleyard, G., A. J. Hapel, and E. A. Boulter. 1971. An antigenic difference between intracellular and extracellular rabbitpox virus. J. Gen. Virol. 13:9-17. - PubMed
-
- Blatch, G. L., and M. Lassle. 1999. The tetratricopeptide repeat: a structural motif mediating protein-protein interactions. Bioessays 21:932-939. - PubMed
-
- Boulanger, D., T. Smith, and M. A. Skinner. 2000. Morphogenesis and release of fowlpox virus. J. Gen. Virol. 81:675-687. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

