Functions of eIF3 downstream of 48S assembly impact AUG recognition and GCN4 translational control
- PMID: 14976554
- PMCID: PMC380973
- DOI: 10.1038/sj.emboj.7600116
Functions of eIF3 downstream of 48S assembly impact AUG recognition and GCN4 translational control
Abstract
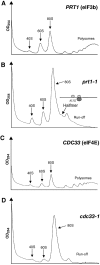
The binding of eIF2-GTP-tRNA(i)(Met) ternary complex (TC) to 40S subunits is impaired in yeast prt1-1 (eIF3b) mutant extracts, but evidence is lacking that TC recruitment is a critical function of eIF3 in vivo. If TC binding was rate-limiting in prt1-1 cells, overexpressing TC should suppress the temperature-sensitive phenotype and GCN4 translation should be strongly derepressed in this mutant, but neither was observed. Rather, GCN4 translation is noninducible in prt1-1 cells, and genetic analysis indicates defective ribosomal scanning between the upstream open reading frames that mediate translational control. prt1-1 cells also show reduced utilization of a near-cognate start codon, implicating eIF3 in AUG selection. Using in vivo cross-linking, we observed accumulation of TC and mRNA/eIF4G on 40S subunits and a 48S 'halfmer' in prt1-1 cells. Genetic evidence suggests that 40S-60S subunit joining is not rate-limiting in the prt1-1 mutant. Thus, eIF3b functions between 48S assembly and subunit joining to influence AUG recognition and reinitiation on GCN4 mRNA. Other mutations that disrupt eIF2-eIF3 contacts in the multifactor complex (MFC) diminished 40S-bound TC, indicating that MFC formation enhances 43S assembly in vivo.
Figures






References
-
- Anand M, Chakraburtty K, Marton MJ, Hinnebusch AG, Kinzy TG (2003) Functional interactions between yeast translation eukaryotic elongation factor (eEF) 1A and eEF3. J Biol Chem 278: 6985–6991 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

