Dissolved oxygen levels alter gene expression and antigen profiles in Borrelia burgdorferi
- PMID: 14977964
- PMCID: PMC356058
- DOI: 10.1128/IAI.72.3.1580-1586.2004
Dissolved oxygen levels alter gene expression and antigen profiles in Borrelia burgdorferi
Erratum in
- Infect Immun. 2004 Apr;72(4):2456
Abstract
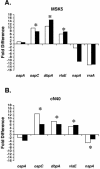
The Lyme disease spirochete, Borrelia burgdorferi, encounters many environmental signals as it cycles between the arthropod vector and mammalian hosts, including temperature, pH, and other host factors. To test the possibility that dissolved oxygen modulates gene expression in B. burgdorferi, spirochetes were exposed to differential levels of dissolved oxygen, and distinct alterations were observed at both the transcriptional and translational levels. Specifically NapA, a Dps/Dpr homologue involved in the oxidative stress response in other bacteria, was reduced when B. burgdorferi was grown under oxygen-limiting conditions. In contrast, several immunoreactive proteins were altered when tested with infection-derived sera from different hosts. Specifically, OspC, DbpA, and VlsE were synthesized at greater levels when cells were grown in limiting oxygen, whereas VraA was reduced. The levels of oxygen in the medium did not affect OspA production. Real-time reverse transcription-PCR analysis of RNA isolated from infectious isolates of strains B31 and cN40 indicated that the expression of ospC, dbpA, and vlsE increased while napA expression decreased under dissolved-oxygen-limiting conditions, whereas flaB was not affected. The reverse transcription-PCR results corroborated the immunoblot analyses and indicated that the increase in OspC, DbpA, and VlsE was due to regulation at the transcriptional level of the genes encoding these antigens. These results indicate that dissolved oxygen modulates gene expression in B. burgdorferi and imply that the redox environment may be an additional regulatory cue that spirochetes exploit to adapt to the disparate niches that they occupy in nature.
Figures



References
-
- Altuvia, S., M. Almiron, G. Huisman, R. Kolter, and G. Storz. 1994. The dps promoter is activated by OxyR during growth and by IHF and sigma S in stationary phase. Mol. Microbiol. 13:265-272. - PubMed
-
- Anguita, J., M. N. Hedrick, and E. Fikrig. 2003. Adaptation of Borrelia burgdorferi in the tick and the mammalian host. FEMS Microbiol. Rev. 27:493-504. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

