Regulated proteolysis by cortical granule serine protease 1 at fertilization
- PMID: 14978210
- PMCID: PMC404006
- DOI: 10.1091/mbc.e03-11-0843
Regulated proteolysis by cortical granule serine protease 1 at fertilization
Abstract
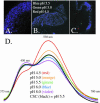
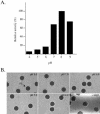
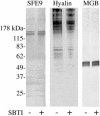
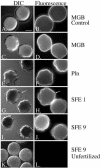
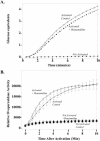
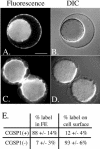
Cortical granules are specialized organelles whose contents interact with the extracellular matrix of the fertilized egg to form the block to polyspermy. In sea urchins, the granule contents form a fertilization envelope (FE), and this construction is critically dependent upon protease activity. An autocatalytic serine protease, cortical granule serine protease 1 (CGSP1), has been identified in the cortical granules of Strongylocentrotus purpuratus eggs, and here we examined the regulation of the protease activity and tested potential target substrates of CGSP1. We found that CGSP1 is stored in its full-length, enzymatically quiescent form in the granule, and is inactive at pH 6.5 or below. We determined the pH of the cortical granule by fluorescent indicators and micro-pH probe measurements and found the granules to be pH 5.5, a condition inhibitory to CGSP1 activity. Exposure of the protease to the pH of seawater (pH 8.0) at exocytosis immediately activates the protease. Activation of eggs at pH 6.5 or lower blocks activation of the protease and the resultant FE phenotypes are indistinguishable from a protease-null phenotype. We find that native cortical granule targets of the protease are beta-1,3 glucanase, ovoperoxidase, and the protease itself, but the structural proteins of the granule are not proteolyzed by CGSP1. Whole mount immunolocalization experiments demonstrate that inhibition of CGSP1 activity affects the localization of ovoperoxidase but does not alter targeting of structural proteins to the FE. The mistargeting of ovoperoxidase may lead to spurious peroxidative cross-linking activity and contribute to the lethality observed in protease-null cells. Thus, CGSP1 is proteolytically active only when secreted, due to the low pH of the cortical granules, and it has a small population of targets for cleavage within the cortical granules.
Figures

Similar articles
-
The cortical granule serine protease CGSP1 of the sea urchin, Strongylocentrotus purpuratus, is autocatalytic and contains a low-density lipoprotein receptor-like domain.Dev Biol. 1999 Jul 1;211(1):1-10. doi: 10.1006/dbio.1999.9299. Dev Biol. 1999. PMID: 10373300
-
Sea urchin ovoperoxidase: oocyte-specific member of a heme-dependent peroxidase superfamily that functions in the block to polyspermy.Mech Dev. 1998 Jan;70(1-2):77-89. doi: 10.1016/s0925-4773(97)00178-0. Mech Dev. 1998. PMID: 9510026
-
Conservation of sequence and function in fertilization of the cortical granule serine protease in echinoderms.Biochem Biophys Res Commun. 2014 Aug 1;450(3):1135-41. doi: 10.1016/j.bbrc.2014.05.098. Epub 2014 May 27. Biochem Biophys Res Commun. 2014. PMID: 24878526 Free PMC article.
-
Knockin' on Egg's Door: Maternal Control of Egg Activation That Influences Cortical Granule Exocytosis in Animal Species.Front Cell Dev Biol. 2021 Sep 3;9:704867. doi: 10.3389/fcell.2021.704867. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34540828 Free PMC article. Review.
-
The biology of cortical granules.Int Rev Cytol. 2001;209:117-206. doi: 10.1016/s0074-7696(01)09012-x. Int Rev Cytol. 2001. PMID: 11580200 Review.
Cited by
-
Cell surface changes in the egg at fertilization.Mol Reprod Dev. 2009 Oct;76(10):942-53. doi: 10.1002/mrd.21090. Mol Reprod Dev. 2009. PMID: 19658159 Free PMC article. Review.
-
Regeneration in bipinnaria larvae of the bat star Patiria miniata induces rapid and broad new gene expression.Mech Dev. 2016 Nov;142:10-21. doi: 10.1016/j.mod.2016.08.003. Epub 2016 Aug 20. Mech Dev. 2016. PMID: 27555501 Free PMC article.
-
Monitoring Proteolytic Activity in Real Time: A New World of Opportunities for Biosensors.Trends Biochem Sci. 2020 Jul;45(7):604-618. doi: 10.1016/j.tibs.2020.03.011. Epub 2020 May 5. Trends Biochem Sci. 2020. PMID: 32386890 Free PMC article. Review.
-
Diversity in the fertilization envelopes of echinoderms.Evol Dev. 2013 Jan;15(1):28-40. doi: 10.1111/ede.12012. Evol Dev. 2013. PMID: 23331915 Free PMC article.
-
A trypsin-like protease with apparent dual function in early Lepeophtheirus salmonis (Krøyer) development.BMC Mol Biol. 2009 May 13;10:44. doi: 10.1186/1471-2199-10-44. BMC Mol Biol. 2009. PMID: 19439101 Free PMC article.
References
-
- Adelson, D.L., and Humphreys, T. (1988). Sea urchin morphogenesis and cell-hyalin adhesion are perturbed by a monoclonal antibody specific for hyalin. Development 104, 391–402. - PubMed
-
- Alliegro, M.C., and Schuel, H. (1985). Characterization of soybean trypsin inhibitor sensitive protease from unfertilized sea urchin eggs. Biochemistry 24, 3926–31. - PubMed
-
- Beschin, A., Bilej, M., Hanssens, F., Raymakers, J., Van Dyck, E., Revets, H., Brys, L., Gomez, J., De Baetselier, P., and Timmermans, M. (1998). Identification and cloning of a glucan- and lipopolysaccharide-binding protein from Eisenia foetida earthworm involved in the activation of prophenoloxidase cascade. J. Biol. Chem. 273, 24948–24954. - PubMed
-
- Bork, P., and Beckmann, G. (1993). The CUB domain. A widespread module in developmentally regulated proteins. J. Mol. Biol. 231, 539–545. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

