Disruption of forkhead transcription factor (FOXO) family members in mice reveals their functional diversification
- PMID: 14978268
- PMCID: PMC365730
- DOI: 10.1073/pnas.0400093101
Disruption of forkhead transcription factor (FOXO) family members in mice reveals their functional diversification
Abstract
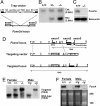
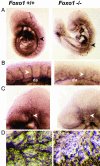
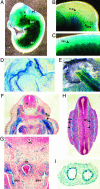
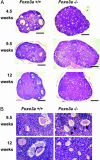
Genetic analysis in Caenorhabditis elegans has uncovered essential roles for DAF-16 in longevity, metabolism, and reproduction. The mammalian orthologs of DAF-16, the closely-related FOXO subclass of forkhead transcription factors (FKHR/FOXO1, FKHRL1/FOXO3a, and AFX/FOXO4), also have important roles in cell cycle arrest, apoptosis and stress responses in vitro, but their in vivo physiological roles are largely unknown. To elucidate their role in normal development and physiology, we disrupted each of the Foxo genes in mice. Foxo1-null embryos died on embryonic day 10.5 as a consequence of incomplete vascular development. Foxo1-null embryonic and yolk sac vessels were not well developed at embryonic day 9.5, and Foxo1 expression was found in a variety of embryonic vessels, suggesting a crucial role of this transcription factor in vascular formation. On the other hand, both Foxo3a- and Foxo4-null mice were viable and grossly indistinguishable from their littermate controls, indicating dispensability of these two members of the Foxo transcription factor family for normal vascular development. Foxo3a-null females showed age-dependent infertility and had abnormal ovarian follicular development. In contrast, histological analyses of Foxo4-null mice did not identify any consistent abnormalities. These results demonstrate that the physiological roles of Foxo genes are functionally diverse in mammals.
Figures

References
-
- Arden, K. C. & Biggs, W. H., III (2002) Arch. Biochem. Biophys. 403, 292-298. - PubMed
-
- Lin, K., Dorman, J. B., Rodan, A. & Kenyon, C. (1997) Science 278, 1319-1322. - PubMed
-
- Ogg, S., Paradis, S., Gottlieb, S., Patterson, G. I., Lee, L., Tissenbaum, H. A. & Ruvkun, G. (1997) Nature 389, 994-999. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

