Dendritic cell aggresome-like induced structures are dedicated areas for ubiquitination and storage of newly synthesized defective proteins
- PMID: 14981091
- PMCID: PMC2172164
- DOI: 10.1083/jcb.200312073
Dendritic cell aggresome-like induced structures are dedicated areas for ubiquitination and storage of newly synthesized defective proteins
Abstract
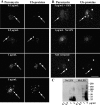
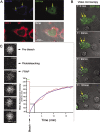
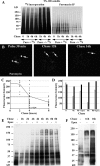
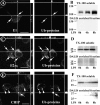
In response to inflammatory stimulation, dendritic cells (DCs) have a remarkable pattern of differentiation (maturation) that exhibits specific mechanisms to control antigen processing and presentation. One of these mechanisms is the sorting of polyubiquitinated proteins in large cytosolic aggregates called dendritic cell aggresome-like induced structures (DALIS). DALIS formation and maintenance are tightly linked to protein synthesis. Here, we took advantage of an antibody recognizing the antibiotic puromycin to follow the fate of improperly translated proteins, also called defective ribosomal products (DRiPs). We demonstrate that DRiPs are rapidly stored and protected from degradation in DALIS. In addition, we show that DALIS contain the ubiquitin-activating enzyme E1, the ubiquitin-conjugating enzyme E225K, and the COOH terminus of Hsp70-interacting protein ubiquitin ligase. The accumulation of these enzymes in the central area of DALIS defines specific functional sites where initial DRiP incorporation and ubiquitination occur. Therefore, DCs are able to regulate DRiP degradation in response to pathogen-associated motifs, a capacity likely to be important for their immune functions.
Figures

References
-
- Banchereau, J., and R.M. Steinman. 1998. Dendritic cells and the control of immunity. Nature. 392:245–252. - PubMed
-
- Chen, Z., and C.M. Pickart. 1990. A 25-kilodalton ubiquitin carrier protein (E2) catalyzes multi-ubiquitin chain synthesis via lysine 48 of ubiquitin. J. Biol. Chem. 265:21835–21842. - PubMed
-
- Chow, A., D. Toomre, W. Garrett, and I. Mellman. 2002. Dendritic cell maturation triggers retrograde MHC class II transport from lysosomes to the plasma membrane. Nature. 418:988–994. - PubMed
-
- Cyr, D.M., J. Hohfeld, and C. Patterson. 2002. Protein quality control: U-box-containing E3 ubiquitin ligases join the fold. Trends Biochem. Sci. 27:368–375. - PubMed
-
- Etlinger, J.D., and A.L. Goldberg. 1980. Control of protein degradation in reticulocytes and reticulocyte extracts by hemin. J. Biol. Chem. 255:4563–4568. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

