Wound-induced HB-EGF ectodomain shedding and EGFR activation in corneal epithelial cells
- PMID: 14985295
- PMCID: PMC2666394
- DOI: 10.1167/iovs.03-0851
Wound-induced HB-EGF ectodomain shedding and EGFR activation in corneal epithelial cells
Abstract
Purpose: Epithelial wound healing is, at least in part, mediated in an autocrine fashion by epidermal growth factor (EGF) receptor (EGFR)-ligand interactions. This study sought to identify the endogenous EGFR ligand and the mechanism by which it is generated in response to wounding in cultured porcine corneas and human corneal epithelial cells.
Methods: Epithelial debridement wounds in cultured porcine corneas and scratch wounds in an epithelial monolayer of SV40-immortalized human corneal epithelial (THCE) cells were allowed to heal in the presence of tyrphostin AG1478 (an EGFR inhibitor), GM6001 (a matrix metalloproteinase [MMP] inhibitor), or CRM197 (a diphtheria toxin mutant), with or without HB-EGF. The activation of EGFR and extracellular signal-regulated kinase (ERK) was analyzed by immunoprecipitation using EGFR antibodies and Western blot analysis with phosphotyrosine antibody. Wound induced HB-EGF shedding was assessed by isolation of secreted HB-EGF from wounded THCE cells and by measuring the release of alkaline phosphatase (AP) in THCE stable cell lines expressing HB-EGF-AP.
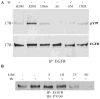
Results: In THCE cells, wound-induced EGFR phosphorylation and ERK activation. In both organ and cell culture models, epithelial wounds were healed in basal media and inhibition of EGFR activation by AG1478 blocked wound closure with or without exogenously added HB-EGF. GM6001 delayed wound closure. Its effects diminished in the presence of exogenous EGF or HB-EGF, suggesting that the MMP inhibitor primarily blocks the release of EGFR ligands. CRM197, a highly specific antagonist of HB-EGF, impaired epithelial wound closure, suggesting that HB-EGF is an endogenous ligand released on epithelial wounding. Consistent with the effects on epithelial migration, all inhibitors as well as HB-EGF function-blocking antibodies retarded wound-induced EGFR phosphorylation in cultured THCE cells. The release of HB-EGF in response to wounding was demonstrated by the fact that heparin-binding proteins isolated from wounded, but not control, THCE-conditioned medium stimulated EGFR and ERK phosphorylation and by the expression of HB-EGF-AP in THCE cells, in which wounding induced the release of AP activity in an MMP-inhibitor-sensitive manner.
Conclusions: HB-EGF released on wounding acts as an autocrine-paracrine EGFR ligand. HB-EGF shedding and EGFR activation represent a critical event during corneal epithelial wound healing, suggesting a possible manipulation of wound healing during the early phases.
Figures

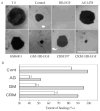
 ) treatments with in the presence of HB-EGF. The coverage of the wound (0% for no migration and 100% for a complete covering of the wound bed) was then calculated. Statistically, there are significant decreases of corneal epithelial healing rate in AG1478-, GM6001-, and CRM197-treated groups compared with MEM-treated (cont, **P < 0.01). Data are the mean ± SE of at least six corneas from two or more independent experiments.
) treatments with in the presence of HB-EGF. The coverage of the wound (0% for no migration and 100% for a complete covering of the wound bed) was then calculated. Statistically, there are significant decreases of corneal epithelial healing rate in AG1478-, GM6001-, and CRM197-treated groups compared with MEM-treated (cont, **P < 0.01). Data are the mean ± SE of at least six corneas from two or more independent experiments.


Similar articles
-
Lysophosphatidic acid promoting corneal epithelial wound healing by transactivation of epidermal growth factor receptor.Invest Ophthalmol Vis Sci. 2007 Feb;48(2):636-43. doi: 10.1167/iovs.06-0203. Invest Ophthalmol Vis Sci. 2007. PMID: 17251460 Free PMC article.
-
ERK1/2 mediate wounding- and G-protein-coupled receptor ligands-induced EGFR activation via regulating ADAM17 and HB-EGF shedding.Invest Ophthalmol Vis Sci. 2009 Jan;50(1):132-9. doi: 10.1167/iovs.08-2246. Epub 2008 Jul 24. Invest Ophthalmol Vis Sci. 2009. PMID: 18658095 Free PMC article.
-
SRC-family tyrosine kinases in wound- and ligand-induced epidermal growth factor receptor activation in human corneal epithelial cells.Invest Ophthalmol Vis Sci. 2006 Jul;47(7):2832-9. doi: 10.1167/iovs.05-1361. Invest Ophthalmol Vis Sci. 2006. PMID: 16799022 Free PMC article.
-
Membrane-anchored growth factors, the epidermal growth factor family: beyond receptor ligands.Cancer Sci. 2008 Feb;99(2):214-20. doi: 10.1111/j.1349-7006.2007.00676.x. Cancer Sci. 2008. PMID: 18271917 Free PMC article. Review.
-
ADAM-mediated ectodomain shedding of HB-EGF in receptor cross-talk.Biochim Biophys Acta. 2005 Aug 1;1751(1):110-7. doi: 10.1016/j.bbapap.2004.11.009. Epub 2004 Dec 8. Biochim Biophys Acta. 2005. PMID: 16054021 Review.
Cited by
-
Free edges in epithelia as cues for motility.Cell Adh Migr. 2011 Mar-Apr;5(2):106-10. doi: 10.4161/cam.5.2.13728. Epub 2011 Mar 1. Cell Adh Migr. 2011. PMID: 20953155 Free PMC article.
-
A toolkit for recombinant production of seven human EGF family growth factors in active conformation.Sci Rep. 2022 Mar 23;12(1):5034. doi: 10.1038/s41598-022-09060-9. Sci Rep. 2022. PMID: 35322149 Free PMC article.
-
EphA2/Ephrin-A1 Mediate Corneal Epithelial Cell Compartmentalization via ADAM10 Regulation of EGFR Signaling.Invest Ophthalmol Vis Sci. 2018 Jan 1;59(1):393-406. doi: 10.1167/iovs.17-22941. Invest Ophthalmol Vis Sci. 2018. PMID: 29351356 Free PMC article.
-
Innate Immune System Activation, Inflammation and Corneal Wound Healing.Int J Mol Sci. 2022 Nov 29;23(23):14933. doi: 10.3390/ijms232314933. Int J Mol Sci. 2022. PMID: 36499260 Free PMC article. Review.
-
Rho kinases regulate corneal epithelial wound healing.Am J Physiol Cell Physiol. 2008 Aug;295(2):C378-87. doi: 10.1152/ajpcell.90624.2007. Epub 2008 May 21. Am J Physiol Cell Physiol. 2008. PMID: 18495812 Free PMC article.
References
-
- Lu L, Reinach PS, Kao WW. Corneal epithelial wound healing. Exp Biol Med. 2001;226:653–664. - PubMed
-
- Gipson I, Sugrue S. Cell Biology of the Corneal Epithelium. In: Albert D, Jakobiec F, editors. Principles and Practice of Ophthalmology. Philadelphia: WB Saunders; 1994. pp. 3–16.
-
- Martin P. Wound healing: aiming for perfect skin regeneration. Science. 1997;276:75–81. - PubMed
-
- Imanishi J, Kamiyama K, Iguchi I, Kita M, Sotozono C, Kinoshita S. Growth factors: importance in wound healing and maintenance of transparency of the cornea. Prog Retin Eye Res. 2000;19:113–129. - PubMed
-
- Wells A. Tumor invasion: role of growth factor-induced cell motility. Adv Cancer Res. 2000;78:31–101. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

