The nitric oxide-guanylyl cyclase signaling pathway modulates membrane activity States and electrophysiological properties of striatal medium spiny neurons recorded in vivo
- PMID: 14985433
- PMCID: PMC6730403
- DOI: 10.1523/JNEUROSCI.4470-03.2004
The nitric oxide-guanylyl cyclase signaling pathway modulates membrane activity States and electrophysiological properties of striatal medium spiny neurons recorded in vivo
Abstract
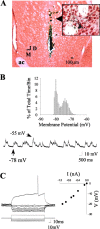
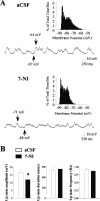
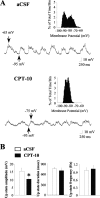
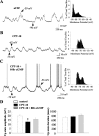
Nitric oxide (NO)-releasing interneurons are believed to regulate the activity of striatal medium spiny neurons (MSNs) that contain the NO effector enzyme guanylyl cyclase (GC). The involvement of NO-GC signaling in modulating steady-state membrane activity of striatal MSNs was examined using in vivo intracellular recordings in rats. Intrastriatal infusion of a neuronal NO synthase inhibitor or a NO scavenger via reverse microdialysis consistently decreased the amplitude of spontaneously occurring depolarized plateau potentials (up events). Intrastriatal infusion of a NO scavenger also decreased the amplitude of EPSPs evoked during electrical stimulation of the orbital prefrontal cortex. The effect of the NO scavenger on spontaneous up events was partially reversed by coperfusion with a cell-permeable cGMP analog. Intracellular injection of MSNs with a soluble GC inhibitor resulted in large decreases in the following: (1) spontaneous up-event amplitude, (2) responsiveness to depolarizing current, (3) action potential amplitude, and (4) input resistance. These effects were partially reversed by coinjection of cGMP. Conversely, intracellular injection of a phosphodiesterase inhibitor increased MSN neuron membrane excitability. These results indicate that, in the intact animal, the NO signaling pathway exerts a powerful tonic modulatory influence over the membrane activity of striatal MSNs via the activation of GC and stimulation of cGMP production.
Figures



Similar articles
-
Nitric oxide-soluble guanylyl cyclase signaling regulates corticostriatal transmission and short-term synaptic plasticity of striatal projection neurons recorded in vivo.Neuropharmacology. 2010 Mar;58(3):624-31. doi: 10.1016/j.neuropharm.2009.11.011. Epub 2009 Dec 5. Neuropharmacology. 2010. PMID: 19969007 Free PMC article.
-
Facilitation of corticostriatal transmission following pharmacological inhibition of striatal phosphodiesterase 10A: role of nitric oxide-soluble guanylyl cyclase-cGMP signaling pathways.J Neurosci. 2015 Apr 8;35(14):5781-91. doi: 10.1523/JNEUROSCI.1238-14.2015. J Neurosci. 2015. PMID: 25855188 Free PMC article.
-
Opposite influences of endogenous dopamine D1 and D2 receptor activation on activity states and electrophysiological properties of striatal neurons: studies combining in vivo intracellular recordings and reverse microdialysis.J Neurosci. 2002 Jan 1;22(1):294-304. doi: 10.1523/JNEUROSCI.22-01-00294.2002. J Neurosci. 2002. PMID: 11756513 Free PMC article.
-
Role of the nitric oxide-soluble guanylyl cyclase pathway in obstructive airway diseases.Pulm Pharmacol Ther. 2014 Oct;29(1):1-6. doi: 10.1016/j.pupt.2014.07.004. Epub 2014 Jul 17. Pulm Pharmacol Ther. 2014. PMID: 25043200 Review.
-
Role of nitric oxide in the regulation of motor function. An overview of behavioral, biochemical and histological studies in animal models.Pharmacol Rep. 2013;65(5):1043-55. doi: 10.1016/s1734-1140(13)71464-6. Pharmacol Rep. 2013. PMID: 24399702 Review.
Cited by
-
7-Nitroindazole reduces L-DOPA-induced dyskinesias in non-human Parkinsonian primate.Open Biol. 2023 May;13(5):220370. doi: 10.1098/rsob.220370. Epub 2023 May 17. Open Biol. 2023. PMID: 37192671 Free PMC article.
-
Targeting Striatal Glutamate and Phosphodiesterases to Control L-DOPA-Induced Dyskinesia.Cells. 2023 Nov 30;12(23):2754. doi: 10.3390/cells12232754. Cells. 2023. PMID: 38067182 Free PMC article. Review.
-
Ischemic-LTP in striatal spiny neurons of both direct and indirect pathway requires the activation of D1-like receptors and NO/soluble guanylate cyclase/cGMP transmission.J Cereb Blood Flow Metab. 2013 Feb;33(2):278-86. doi: 10.1038/jcbfm.2012.167. Epub 2012 Nov 14. J Cereb Blood Flow Metab. 2013. PMID: 23149555 Free PMC article.
-
Is there a role for nitric oxide in methamphetamine-induced dopamine terminal degeneration?Neurotox Res. 2014 Feb;25(2):153-60. doi: 10.1007/s12640-013-9415-2. Epub 2013 Aug 6. Neurotox Res. 2014. PMID: 23918001 Free PMC article. Review.
-
Concepts of neural nitric oxide-mediated transmission.Eur J Neurosci. 2008 Jun;27(11):2783-802. doi: 10.1111/j.1460-9568.2008.06285.x. Eur J Neurosci. 2008. PMID: 18588525 Free PMC article. Review.
References
-
- Ariano MA (1983) Distribution of the components of the guanosine 3′,5′-phosphate system in rat caudate-putamen. Neuroscience 10: 707-723. - PubMed
-
- Bockelmann R, Wolf G, Ransmayr G, Riederer P (1994) NADPH-diaphorase/nitric oxide synthase containing in normal and Parkinson's disease putamen. J Neural Transm Park Dis Dement Sect 7: 115-121. - PubMed
-
- Calabresi P, Centonze D, Gubellini P, Marfia GA, Pisani A, Sancesario G, Bernardi G (2000a) Synaptic transmission in the striatum: from plasticity to neurodegeneration. Prog Neurobiol 61: 231-265. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
