Myosin V: regulation by calcium, calmodulin, and the tail domain
- PMID: 15007063
- PMCID: PMC2172279
- DOI: 10.1083/jcb.200310065
Myosin V: regulation by calcium, calmodulin, and the tail domain
Abstract
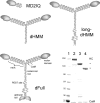
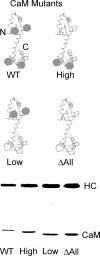
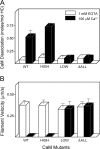
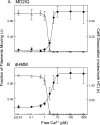
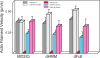
Calcium activates the ATPase activity of tissue-purified myosin V, but not that of shorter expressed constructs. Here, we resolve this discrepancy by comparing an expressed full-length myosin V (dFull) to three shorter constructs. Only dFull has low ATPase activity in EGTA, and significantly higher activity in calcium. Based on hydrodynamic data and electron microscopic images, the inhibited state is due to a compact conformation that is possible only with the whole molecule. The paradoxical finding that dFull moved actin in EGTA suggests that binding of the molecule to the substratum turns it on, perhaps mimicking cargo activation. Calcium slows, but does not stop the rate of actin movement if excess calmodulin (CaM) is present. Without excess CaM, calcium binding to the high affinity sites dissociates CaM and stops motility. We propose that a folded-to-extended conformational change that is controlled by calcium and CaM, and probably by cargo binding itself, regulates myosin V's ability to transport cargo in the cell.
Figures



References
-
- Beckingham, K. 1991. Use of site-directed mutations in the individual Ca2+-binding sites of calmodulin to examine Ca2+-induced conformational changes. J. Biol. Chem. 266:6027–6030. - PubMed
-
- Casaletti, L., S.B. Tauhata, J.E. Moreira, and R.E. Larson. 2003. Myosin-Va proteolysis by Ca2+/calpain in depolarized nerve endings from rat brain. Biochem. Biophys. Res. Commun. 308:159–164. - PubMed
-
- Cheney, R.E., M.K. O'Shea, J.E. Heuser, M.V. Coelho, J.S. Wolenski, E.M. Espreafico, P. Forscher, R.E. Larson, and M.S. Mooseker. 1993. Brain myosin-V is a two-headed unconventional myosin with motor activity. Cell. 75:13–23. - PubMed
-
- Coy, D.L., W.O. Hancock, M. Wagenbach, and J. Howard. 1999. Kinesin's tail domain is an inhibitory regulator of the motor domain. Nat. Cell Biol. 1:288–292. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

