The actions of monoamines and distribution of noradrenergic and serotoninergic contacts on different subpopulations of commissural interneurons in the cat spinal cord
- PMID: 15016088
- PMCID: PMC1971244
- DOI: 10.1111/j.1460-9568.2004.03239.x
The actions of monoamines and distribution of noradrenergic and serotoninergic contacts on different subpopulations of commissural interneurons in the cat spinal cord
Abstract
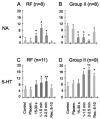
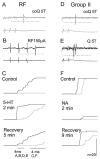
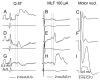
Modulatory actions of monoamines were investigated on spinal commissural interneurons which coordinate left-right hindlimb muscle activity through direct projections to the contralateral motor nuclei. Commissural interneurons located in Rexed lamina VIII, with identified projections to the contralateral gastrocnemius-soleus motor nuclei, were investigated in deeply anaesthetized cats. Most interneurons had dominant input from either the reticular formation or from group II muscle afferents; a small proportion of neurons had input from both. Actions of ionophoretically applied serotonin and noradrenaline were examined on extracellularly recorded spikes evoked monosynaptically by group II muscle afferents or reticulospinal tract fibres. Activation by reticulospinal fibres was facilitated by both serotonin and noradrenaline. Activation by group II afferents was also facilitated by serotonin but was strongly depressed by noradrenaline. To investigate the possible morphological substrates of this differential modulation, seven representative commissural interneurons were labelled intracellularly with tetramethylrhodamine-dextran and neurobiotin. Contacts from noradrenergic and serotoninergic fibres were revealed by immunohistochemistry and analysed with confocal microscopy. There were no major differences in the numbers and distributions of contacts among the interneurons studied. The findings suggest that differences in modulatory actions of monoamines, and subsequent changes in the recruitment of subpopulations of commissural interneurons in various behavioural situations, depend on intrinsic interneuron properties rather than on the patterns of innervation by monoaminergic fibres. The different actions of noradrenaline on different populations of interneurons might permit reconfiguration of the actions of the commissural neurons according to behavioural context.
Figures




Similar articles
-
Differential modulation by monoamine membrane receptor agonists of reticulospinal input to lamina VIII feline spinal commissural interneurons.Eur J Neurosci. 2007 Sep;26(5):1205-12. doi: 10.1111/j.1460-9568.2007.05764.x. Eur J Neurosci. 2007. PMID: 17767499
-
Serotoninergic and noradrenergic axonal contacts associated with premotor interneurons in spinal pathways from group II muscle afferents.Eur J Neurosci. 2000 Apr;12(4):1271-80. doi: 10.1046/j.1460-9568.2000.00022.x. Eur J Neurosci. 2000. PMID: 10762356
-
Commissural interneurons with input from group I and II muscle afferents in feline lumbar segments: neurotransmitters, projections and target cells.J Physiol. 2009 Jan 15;587(2):401-18. doi: 10.1113/jphysiol.2008.159236. Epub 2008 Dec 1. J Physiol. 2009. PMID: 19047210 Free PMC article.
-
The mammalian spinal commissural system: properties and functions.J Neurophysiol. 2020 Jan 1;123(1):4-21. doi: 10.1152/jn.00347.2019. Epub 2019 Nov 6. J Neurophysiol. 2020. PMID: 31693445 Free PMC article. Review.
-
Modulation of information forwarded to feline cerebellum by monoamines.Ann N Y Acad Sci. 1998 Nov 16;860:106-9. doi: 10.1111/j.1749-6632.1998.tb09042.x. Ann N Y Acad Sci. 1998. PMID: 9928305 Review.
Cited by
-
Segmental, synaptic actions of commissural interneurons in the mouse spinal cord.J Neurosci. 2007 Jun 13;27(24):6521-30. doi: 10.1523/JNEUROSCI.1618-07.2007. J Neurosci. 2007. PMID: 17567813 Free PMC article.
-
Systematic Morphometry of Catecholamine Nuclei in the Brainstem.Front Neuroanat. 2017 Nov 2;11:98. doi: 10.3389/fnana.2017.00098. eCollection 2017. Front Neuroanat. 2017. PMID: 29163071 Free PMC article.
-
Functional differentiation and organization of feline midlumbar commissural interneurones.J Physiol. 2005 Jun 1;565(Pt 2):645-58. doi: 10.1113/jphysiol.2005.083014. Epub 2005 Apr 7. J Physiol. 2005. PMID: 15817636 Free PMC article.
-
Role of Descending Serotonergic Fibers in the Development of Pathophysiology after Spinal Cord Injury (SCI): Contribution to Chronic Pain, Spasticity, and Autonomic Dysreflexia.Biology (Basel). 2022 Feb 1;11(2):234. doi: 10.3390/biology11020234. Biology (Basel). 2022. PMID: 35205100 Free PMC article. Review.
-
Modelling genetic reorganization in the mouse spinal cord affecting left-right coordination during locomotion.J Physiol. 2013 Nov 15;591(22):5491-508. doi: 10.1113/jphysiol.2013.261115. Epub 2013 Sep 30. J Physiol. 2013. PMID: 24081162 Free PMC article.
References
-
- Aggelopoulos NC, Edgley SA. Segmental localisation of the relays mediating crossed inhibition of hindlimb motoneurones from group II afferents in the anaesthetized cat spinal cord. Neurosci Lett. 1995;185:60–64. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

