Primary central nervous system lymphoma expressing the human neurotropic polyomavirus, JC virus, genome
- PMID: 15016869
- PMCID: PMC371040
- DOI: 10.1128/jvi.78.7.3462-3469.2004
Primary central nervous system lymphoma expressing the human neurotropic polyomavirus, JC virus, genome
Abstract
B lymphocytes are known as a potential site for latency and reactivation of the human neurotropic polyomavirus, JC virus (JCV). In light of recent studies on the oncogenicity of JCV and the transforming ability of the JCV early protein, T antigen, we investigated the association of JCV with B-cell lymphomas of the central nervous system. Examination of 27 well-characterized clinical specimens by gene amplification and immunohistochemistry revealed the presence of DNA sequences corresponding to the JCV early genome and the late Agnoprotein in 22 samples and the JCV late genome encoding the viral capsid proteins in 8 samples. Expression of T antigen and that of Agnoprotein by immunohistochemistry were each detected in six specimens. No evidence of the production of viral capsid proteins was observed, ruling out productive infection of JCV in the tumor cells. The results from laser capture microdissection verified the presence of JCV T-antigen sequences in tumor cells with positive immunoreactivity to antibodies against the viral proteins T antigen and Agnoprotein. Due to previous reports demonstrating an association of the Epstein-Barr virus (EBV) with transformation of B lymphocytes, EBV DNA sequences and the EBV transforming protein, latent membrane protein 1 (LMP1), were analyzed in parallel. EBV LMP1 DNA sequences were detected in 16 of 23 samples, and LMP1 expression was detected in 16 samples, 5 of which exhibited positive immunoreactivity to JCV proteins. Double labeling demonstrated coexpression of JCV T antigen and EBV LMP1 in the same cells. The detection of the JCV genome in large numbers of B-cell lymphomas and its coexistence with EBV suggest a potential role for JCV in the pathogenesis of primary CNS lymphoma.
Figures


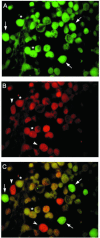
Similar articles
-
The Role of the JC Virus in Central Nervous System Tumorigenesis.Int J Mol Sci. 2020 Aug 28;21(17):6236. doi: 10.3390/ijms21176236. Int J Mol Sci. 2020. PMID: 32872288 Free PMC article. Review.
-
Detection of JC virus DNA sequences and expression of viral T antigen and agnoprotein in esophageal carcinoma.Cancer. 2005 Feb 1;103(3):516-27. doi: 10.1002/cncr.20806. Cancer. 2005. PMID: 15630684
-
Expression of human neurotropic polyomavirus JCV late gene product agnoprotein in human medulloblastoma.J Natl Cancer Inst. 2002 Feb 20;94(4):267-73. doi: 10.1093/jnci/94.4.267. J Natl Cancer Inst. 2002. PMID: 11854388
-
Prevalence of JC polyomavirus large T antigen sequences among Iranian patients with central nervous system tumors.Arch Virol. 2015 Jan;160(1):61-8. doi: 10.1007/s00705-014-2230-0. Epub 2014 Sep 14. Arch Virol. 2015. PMID: 25218012
-
Potential mechanisms of the human polyomavirus JC in neural oncogenesis.J Neuropathol Exp Neurol. 2008 Aug;67(8):729-40. doi: 10.1097/NEN.0b013e318180e631. J Neuropathol Exp Neurol. 2008. PMID: 18648329 Free PMC article. Review.
Cited by
-
Molecular biology, epidemiology, and pathogenesis of progressive multifocal leukoencephalopathy, the JC virus-induced demyelinating disease of the human brain.Clin Microbiol Rev. 2012 Jul;25(3):471-506. doi: 10.1128/CMR.05031-11. Clin Microbiol Rev. 2012. PMID: 22763635 Free PMC article. Review.
-
The Role of the JC Virus in Central Nervous System Tumorigenesis.Int J Mol Sci. 2020 Aug 28;21(17):6236. doi: 10.3390/ijms21176236. Int J Mol Sci. 2020. PMID: 32872288 Free PMC article. Review.
-
High JC virus load in gastric cancer and adjacent non-cancerous mucosa.Cancer Sci. 2007 Jan;98(1):25-31. doi: 10.1111/j.1349-7006.2006.00354.x. Cancer Sci. 2007. PMID: 17083566 Free PMC article.
-
Human Polyomavirus JCPyV and Its Role in Progressive Multifocal Leukoencephalopathy and Oncogenesis.Front Oncol. 2019 Aug 8;9:711. doi: 10.3389/fonc.2019.00711. eCollection 2019. Front Oncol. 2019. PMID: 31440465 Free PMC article. Review.
-
Opportunistic DNA Recombination With Epstein-Barr Virus at Sites of Control Region Rearrangements Mediating JC Virus Neurovirulence.J Infect Dis. 2016 May 1;213(9):1436-43. doi: 10.1093/infdis/jiv755. Epub 2015 Dec 21. J Infect Dis. 2016. PMID: 26690342 Free PMC article.
References
-
- Andreoletti, L., V. Dubois, A. Lescieux, A. Dewilde, L. Bocket, H. J. Fleury, and P. Wattre. 1999. Human polyomavirus JC latency and reactivation status in blood of HIV-1 positive immunocompromised patients with and without progressive multifocal leukoencephalopathy. AIDS 13:1469-1475. - PubMed
-
- Andreoletti, L., A. Lescieux, V. Lambert, A. Si-Mohamed, M. Matta, P. Wattre, and L. Belec. 2002. Semiquantitative detection of JCV DNA in peripheral blood leukocytes from HIV-1-infected patients with or without progressive multifocal leukoencephalopathy. J. Med. Virol. 66:1-7. - PubMed
-
- Astrom, K. E., E. L. Mancall, and E. P. Richardson, Jr. 1958. Progressive multifocal leukoencephalopathy: a hitherto unrecognized complication of chronic lymphatic leukemia and Hodgkin's disease. Brain 81:93-111. - PubMed
-
- Atwood, W. J., K. Amemiya, R. Traub, J. Harms, and E. O. Major. 1992. Interaction of the human polyomavirus, JCV, with human B-lymphocytes. Virology 190:716-723. - PubMed
-
- Berger, J. R., and M. Concha. 1995. Progressive multifocal leukoencephalopathy: the evolution of a disease once considered rare. J. Neurovirol. 1:15-18. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

