Mitosis-specific hyperphosphorylation of Epstein-Barr virus nuclear antigen 2 suppresses its function
- PMID: 15016877
- PMCID: PMC371044
- DOI: 10.1128/jvi.78.7.3542-3552.2004
Mitosis-specific hyperphosphorylation of Epstein-Barr virus nuclear antigen 2 suppresses its function
Abstract
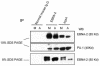
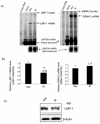
The Epstein-Barr virus (EBV) nuclear antigen 2 (EBNA-2) is a key gene expressed in EBV type III latent infection that can transactivate numerous promoters, including those for all the other type III viral latency genes as well as cellular genes responsible for cell proliferation. EBNA-2 is essential for EBV-mediated immortalization of primary B lymphocytes. We now report that EBNA-2, a phosphoprotein, is hyperphosphorylated specifically in mitosis. Evidence that the cyclin-dependent kinase p34(cdc2) may be involved in this hyperphosphorylation includes (i) coimmunoprecipitation of EBNA-2 and p34(cdc2), suggesting physical association; (ii) temporal correlation between hyperphosphorylation of EBNA-2 and an increase in p34(cdc2) kinase activity; and (iii) ability of purified p34(cdc2)/cyclin B1 kinase to phosphorylate EBNA-2 in vitro. Hyperphosphorylation of EBNA-2 appears to suppress its ability to transactivate the latent membrane protein 1 (LMP-1) promoter by about 50%. The association between EBNA-2 and PU.1 is also decreased by about 50% in M-phase-arrested cells, as shown by coimmunoprecipitation from cell lysates, suggesting that hyperphosphorylation of EBNA-2 impairs its affinity for PU.1. Finally, endogenous LMP-1 mRNA levels in M phase are around 55% of those in asynchronously growing cells. These results suggest that regulation of gene expression during type III latency may be regulated in a cell-cycle-related manner.
Figures





Similar articles
-
cdc2/cyclin B1-dependent phosphorylation of EBNA2 at Ser243 regulates its function in mitosis.J Virol. 2006 Feb;80(4):2045-50. doi: 10.1128/JVI.80.4.2045-2050.2006. J Virol. 2006. PMID: 16439560 Free PMC article.
-
Epstein-barr virus nuclear antigen 3C activates the latent membrane protein 1 promoter in the presence of Epstein-Barr virus nuclear antigen 2 through sequences encompassing an spi-1/Spi-B binding site.J Virol. 2000 Jun;74(11):5151-60. doi: 10.1128/jvi.74.11.5151-5160.2000. J Virol. 2000. PMID: 10799590 Free PMC article.
-
Interferon regulatory factor 7 is induced by Epstein-Barr virus latent membrane protein 1.J Virol. 2000 Feb;74(3):1061-8. doi: 10.1128/jvi.74.3.1061-1068.2000. J Virol. 2000. PMID: 10627515 Free PMC article.
-
Epstein-Barr virus latent genes.Exp Mol Med. 2015 Jan 23;47(1):e131. doi: 10.1038/emm.2014.84. Exp Mol Med. 2015. PMID: 25613728 Free PMC article. Review.
-
Cytokine mediated induction of the major Epstein-Barr virus (EBV)-encoded transforming protein, LMP-1.Immunol Lett. 2006 Apr 15;104(1-2):83-8. doi: 10.1016/j.imlet.2005.11.003. Epub 2005 Dec 1. Immunol Lett. 2006. PMID: 16386314 Review.
Cited by
-
Species-specific functions of Epstein-Barr virus nuclear antigen 2 (EBNA2) reveal dual roles for initiation and maintenance of B cell immortalization.PLoS Pathog. 2017 Dec 20;13(12):e1006772. doi: 10.1371/journal.ppat.1006772. eCollection 2017 Dec. PLoS Pathog. 2017. PMID: 29261800 Free PMC article.
-
Functional diversity: update of the posttranslational modification of Epstein-Barr virus coding proteins.Cell Mol Life Sci. 2022 Nov 14;79(12):590. doi: 10.1007/s00018-022-04561-2. Cell Mol Life Sci. 2022. PMID: 36376593 Free PMC article. Review.
-
cdc2/cyclin B1-dependent phosphorylation of EBNA2 at Ser243 regulates its function in mitosis.J Virol. 2006 Feb;80(4):2045-50. doi: 10.1128/JVI.80.4.2045-2050.2006. J Virol. 2006. PMID: 16439560 Free PMC article.
-
RNA polymerase II stalling promotes nucleosome occlusion and pTEFb recruitment to drive immortalization by Epstein-Barr virus.PLoS Pathog. 2011 Oct;7(10):e1002334. doi: 10.1371/journal.ppat.1002334. Epub 2011 Oct 27. PLoS Pathog. 2011. PMID: 22046134 Free PMC article.
-
Epstein-Barr virus protein kinase BGLF4 is a virion tegument protein that dissociates from virions in a phosphorylation-dependent process and phosphorylates the viral immediate-early protein BZLF1.J Virol. 2006 Jun;80(11):5125-34. doi: 10.1128/JVI.02674-05. J Virol. 2006. PMID: 16698993 Free PMC article.
References
-
- Ben-Bassat, H., N. Goldblum, S. Mitrani, T. Goldblum, J. M. Yoffey, M. M. Cohen, Z. Bentwith, B. Ramot, E. Klein, and G. Klein. 1977. Establishment in continuous culture of a new type of lymphocyte from a “Burkitt-like” malignant lymphoma (line D.G.-75). Int. J. Cancer 19:27-33. - PubMed
-
- Conteras-Brodin, B. A., M. Anvret, S. Imreh, E. Altiok, G. Klein, and M. G. Masucci. 1991. B cell phenotype-dependent expression of the Epstein-Barr virus nuclear antigens EBNA-2 to EBNA-6: studies with somatic cell hybrids. J. Gen. Virol. 72:3025-3033. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

