Analytic models for mechanotransduction: gating a mechanosensitive channel
- PMID: 15024097
- PMCID: PMC384697
- DOI: 10.1073/pnas.0307804101
Analytic models for mechanotransduction: gating a mechanosensitive channel
Abstract
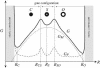
Analytic estimates for the forces and free energy generated by bilayer deformation reveal a compelling and intuitive model for MscL channel gating analogous to the nucleation of a second phase. We argue that the competition between hydrophobic mismatch and tension results in a surprisingly rich story that can provide both a quantitative comparison with measurements of opening tension for MscL when reconstituted in bilayers of different thickness, and qualitative insights into the function of the MscL channel and other transmembrane proteins.
Figures



References
-
- Blount, P. & Moe, P. C. (1999) Trends Microbiol. 7, 420–424. - PubMed
-
- Gillespie, P. G. & Walker, R. G. (2001) Nature 413, 194–202. - PubMed
-
- Sachs, F. (1992) Soc. Gen. Physiol. Ser. 47, 241–260. - PubMed
-
- Perozo, E., Kloda, A., Marien Cortes, D. & Martinac, B. (2002) Nat. Struct. Biol. 9, 696–703. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

