Circadian and diurnal calcium oscillations encode photoperiodic information in Arabidopsis
- PMID: 15031410
- PMCID: PMC412869
- DOI: 10.1105/tpc.020214
Circadian and diurnal calcium oscillations encode photoperiodic information in Arabidopsis
Abstract
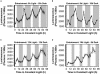
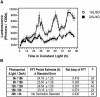
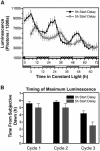
We have tested the hypothesis that circadian oscillations in the concentration of cytosolic free calcium ([Ca2+]cyt) can encode information. We imaged oscillations of [Ca2+]cyt in the cotyledons and leaves of Arabidopsis (Arabidopsis thaliana) that have a 24-h period in light/dark cycles and also constant light. The amplitude, phase, and shape of the oscillations of [Ca2+]cyt and [Ca2+]cyt at critical daily time points were controlled by the light/dark regimes in which the plants were grown. These data provide evidence that 24-h oscillations in [Ca2+]cyt encode information concerning daylength and light intensity, which are two major regulators of plant growth and development.
Figures




Similar articles
-
Circadian oscillations of cytosolic and chloroplastic free calcium in plants.Science. 1995 Sep 29;269(5232):1863-5. doi: 10.1126/science.7569925. Science. 1995. PMID: 7569925
-
Time of day modulates low-temperature Ca signals in Arabidopsis.Plant J. 2006 Dec;48(6):962-73. doi: 10.1111/j.1365-313X.2006.02933.x. Plant J. 2006. PMID: 17227550
-
Coupling diurnal cytosolic Ca2+ oscillations to the CAS-IP3 pathway in Arabidopsis.Science. 2007 Mar 9;315(5817):1423-6. doi: 10.1126/science.1134457. Science. 2007. PMID: 17347443
-
Coordination of plastid and light signaling pathways upon development of Arabidopsis leaves under various photoperiods.Mol Plant. 2012 Jul;5(4):799-816. doi: 10.1093/mp/ssr106. Epub 2011 Dec 23. Mol Plant. 2012. PMID: 22199239 Free PMC article. Review.
-
From a repressilator-based circadian clock mechanism to an external coincidence model responsible for photoperiod and temperature control of plant architecture in Arabodopsis thaliana.Biosci Biotechnol Biochem. 2013;77(1):10-6. doi: 10.1271/bbb.120765. Epub 2013 Jan 7. Biosci Biotechnol Biochem. 2013. PMID: 23291766 Review.
Cited by
-
The RON1/FRY1/SAL1 gene is required for leaf morphogenesis and venation patterning in Arabidopsis.Plant Physiol. 2010 Mar;152(3):1357-72. doi: 10.1104/pp.109.149369. Epub 2009 Dec 31. Plant Physiol. 2010. PMID: 20044451 Free PMC article.
-
Circadian oscillations of cytosolic free calcium regulate the Arabidopsis circadian clock.Nat Plants. 2018 Sep;4(9):690-698. doi: 10.1038/s41477-018-0224-8. Epub 2018 Aug 20. Nat Plants. 2018. PMID: 30127410 Free PMC article.
-
CML24, regulated in expression by diverse stimuli, encodes a potential Ca2+ sensor that functions in responses to abscisic acid, daylength, and ion stress.Plant Physiol. 2005 Sep;139(1):240-53. doi: 10.1104/pp.105.062612. Epub 2005 Aug 19. Plant Physiol. 2005. PMID: 16113225 Free PMC article.
-
Dynamics of leaf and root growth: endogenous control versus environmental impact.Ann Bot. 2005 May;95(6):891-900. doi: 10.1093/aob/mci103. Epub 2005 Mar 14. Ann Bot. 2005. PMID: 15767269 Free PMC article. Review.
-
Cellular Calcium Levels Influenced by NCA-2 Impact Circadian Period Determination in Neurospora.mBio. 2021 Jun 29;12(3):e0149321. doi: 10.1128/mBio.01493-21. Epub 2021 Jun 29. mBio. 2021. PMID: 34182778 Free PMC article.
References
-
- Alabadi, D., Oyama, T., Yanovsky, M.J., Harmon, F.G., Mas, P., and Kay, S.A. (2001). Reciprocal regulation between TOC1 and LHY/CCA1 within the Arabidopsis circadian clock. Science 293, 880–883. - PubMed
-
- Allen, G.J., Chu, S.P., Harrington, C.L., Schumacher, K., Hoffmann, T., Tang, Y.Y., Grill, E., and Schroeder, J.I. (2001). A defined range of guard cell calcium oscillation parameters encodes stomatal movements. Nature 411, 1053–1057. - PubMed
-
- Bakrim, N., Brulfert, J., Vidal, J., and Chollet, R. (2001). Phosphoenolpyruvate carboxylase kinase is controlled by a similar signaling cascade in CAM and C4 plants. Biochem. Biophys. Res. Commun. 286, 1158–1162. - PubMed
-
- Berridge, M.J., Lipp, P., and Bootman, M.D. (2000). The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 1, 11–21. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

