Pathology affects different organs in two mouse strains chronically infected by a Trypanosoma cruzi clone: a model for genetic studies of Chagas' disease
- PMID: 15039360
- PMCID: PMC375186
- DOI: 10.1128/IAI.72.4.2350-2357.2004
Pathology affects different organs in two mouse strains chronically infected by a Trypanosoma cruzi clone: a model for genetic studies of Chagas' disease
Abstract
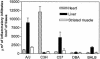
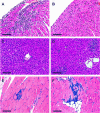
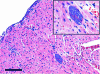
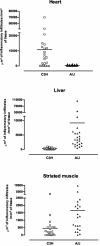
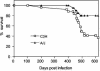
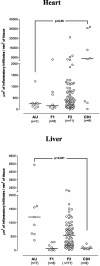
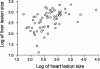
Chagas' disease is a chronic infection caused by Trypanosoma cruzi and represents an important public health burden in Latin America. Frequently the disease evolves undetectable for decades, while in a significant fraction of the affected individuals it culminates in death by heart failure. Here, we describe a novel murine model of the chronic infection with T. cruzi using a stable clone isolated from a human patient (Sylvio X10/4). The infection in the C3H/HePAS mouse strain progresses chronically and is mainly characterized by intense cardiac inflammatory lesions that recapitulate the chronic cardiac pathology observed in the human disease. Moderate striated muscle lesions are also present in C3H/HePAS mice. Viable parasites are detected and recovered from the chronic heart lesions of C3H/HePAS mice, supporting the current notion that development of heart pathology in Chagas' disease is related to parasite persistence in the inflamed tissue. By contrast, in infected A/J mice, chronic inflammatory lesions are targeted to the liver and the skeletal muscle, while pathology and parasites are undetectable in the heart. The phenotypic analysis of F(1) (A/J x C3H/HePAS) and F(2) (A/J x C3H/HePAS) mice suggests that the genetic predisposition to develop the inflammatory lesions caused by T. cruzi (Sylvio X10/4 clone) is heterogeneous because the heart and liver pathology segregate in the F(2) generation. These findings raise the hypothesis that the pathology heterogeneity observed in humans with Chagas' disease (absence and presence of cardiac or digestive chronic lesions) may be attributable to host genetic factors.
Figures

References
-
- Andrade, L. O., C. R. Machado, E. Chiari, S. D. Pena, and A. M. Macedo. 1999. Differential tissue distribution of diverse clones of Trypanosoma cruzi in infected mice. Mol. Biochem. Parasitol. 100:163-172. - PubMed
-
- Andrade, S. G., and Z. A. Andrade. 1968. Pathology of prolonged experimental Chagas’ disease. Rev. Inst. Med. Trop. Sao Paulo 10:180-187. - PubMed
-
- Anez, N., H. Carrasco, H. Parada, G. Crisante, A. Rojas, C. Fuenmayor, N. Gonzalez, G. Percoco, R. Borges, P. Guevara, and J. L. Ramirez. 1999. Myocardial parasite persistence in chronic chagasic patients. Am. J. Trop. Med. Hyg. 60:726-732. - PubMed
-
- Cunha-Neto, E., V. Coelho, L. Guilherme, A. Fiorelli, N. Stolf, and J. Kalil. 1996. Autoimmunity in Chagas’ disease. Identification of cardiac myosin-B13 Trypanosoma cruzi protein crossreactive T cell clones in heart lesions of a chronic Chagas’ cardiomyopathy patient. J. Clin. Investig. 98:1709-1712. - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

