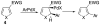Palladium-catalyzed arylation and heteroarylation of indolizines
- PMID: 15040747
- PMCID: PMC3708550
- DOI: 10.1021/ol049866q
Palladium-catalyzed arylation and heteroarylation of indolizines
Abstract
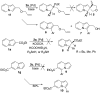
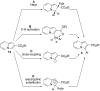
A highly effective protocol for palladium-catalyzed selective arylation and heteroarylation of indolizines at C-3 has been developed. Mechanistic studies unambiguously support an electrophilic substitution pathway for this transformation.
Figures
References
-
-
For recent examples, see: Sawada K, Okada S, Kuroda A, Watanabe S, Sawada Y, Tanaka H. Chem Pharm Bull. 2001;49:799.Ostby OB, Dalhus B, Gundersen LL, Rise F, Bast A, Haenen GRMM. Eur J Org Chem. 2000;22:3763.
-
-
-
For recent reviews, see: Michael JP. Alkaloids. 2001;55:91.Michael JP. Nat Prod Rep. 2002;19:742.
-
-
-
See, for example: Halab L, Becker JAJ, Darula Z, Tourwe D, Kieffer BL, Simonin F, Lubell WD. J Med Chem. 2002;45:5353.Pearson WH, Hembre EJ. J Org Chem. 1996;61:5546.
-
-
-
For a review on syntheses and properties of indolizines, see: Behnisch A, Behnisch P, Eggenweiler M, Wallenhorst T Indolizine. Houben-Weyl. 1994;E6b/1, 2a:323–450.
-
-
-
For original papers, see: Chichibabin AE. Chem Ber. 1927;60B:1607.Chichibabin AE, Stepanov FN. Chem Ber. 1929;62B:1068.
-
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous