Block of long-term potentiation by naturally secreted and synthetic amyloid beta-peptide in hippocampal slices is mediated via activation of the kinases c-Jun N-terminal kinase, cyclin-dependent kinase 5, and p38 mitogen-activated protein kinase as well as metabotropic glutamate receptor type 5
- PMID: 15056716
- PMCID: PMC6730034
- DOI: 10.1523/JNEUROSCI.1633-03.2004
Block of long-term potentiation by naturally secreted and synthetic amyloid beta-peptide in hippocampal slices is mediated via activation of the kinases c-Jun N-terminal kinase, cyclin-dependent kinase 5, and p38 mitogen-activated protein kinase as well as metabotropic glutamate receptor type 5
Abstract
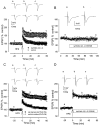
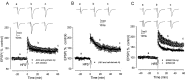
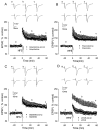
The mechanisms of action of human synthetic and naturally secreted cell-derived amyloid beta-peptide (Abeta)(1-42) on the induction of long-term potentiation (LTP) were investigated in the medial perforant path to dentate granule cell synapses in hippocampal slices. Synthetic and cell-derived Abeta strongly inhibited high-frequency stimulation (HFS)-induced LTP at peak HFS and 1 hr after HFS. Cell-derived Abeta was much more potent than synthetic Abeta at inhibiting LTP induction, with threshold concentrations of approximately 1 and 100-200 nm, respectively. The involvement of various kinases in Abeta-mediated inhibition of LTP induction was investigated by applying Abeta in the presence of inhibitors of these kinases. The c-Jun N-terminal kinase (JNK) inhibitor JNKI prevented the block of LTP induction by both synthetic and cell-derived Abeta. The block of LTP induced by synthetic Abeta was also prevented by the JNK inhibitor anthra[1,9-cd]pyrazol-6(2H)-one, the cyclin-dependent kinase 5 (Cdk5) inhibitors butyrolactone and roscovitine, and the p38 MAP kinase (MAPK) inhibitor 4-(4-fluorophenyl)-2-(4-methylsulfonylphenyl)-5-(4-pyridyl)-1H-imidazole but not by the p42-p44 MAP kinase inhibitor 1,4-diamino-2,3-dicyano-1,4-bis(2-aminophenylthio)butadiene. The group I-group II metabotropic glutamate receptor (mGluR) antagonist 2S-2-amino-2-(1S,2S-2-carboxycyclopropyl-1-yl)-3-(xanth-9-yl)propanoic acid and the mGluR5 antagonist methyl-6-(phenylethynyl)pyridine prevented the block of LTP induction by Abeta. However, thealpha7 nicotinic ACh receptor antagonist methylcaconatine did not prevent the inhibition of LTP induction by Abeta. These studies provide evidence that the Abeta-mediated inhibition of LTP induction involves stimulation of the kinases JNK, Cdk5, and p38 MAPK after the activation of both the Abeta receptor(s) and mGluR5.
Figures

References
-
- Ahlijanian MK, Barrezueta NX, Williams RD, Jakowski A, Kowsz KP, McCarthy S, Coskran T, Carlo A, Seymour PA, Burkhardt JE, Nelson RB, McNeish JD (2000) Hyperphosphorylated tau and neurofilament and cytoskeletal disruptions in mice overexpressing human p25, an activator of Cdk5. Proc Natl Acad Sci USA 97: 2910–2915. - PMC - PubMed
-
- Akama KT, Van Eldick L (2000) β-Amyloid stimulation of inducible nitric oxide synthase in astrocytes is interleukin-1-β and tumor necrosis factor-α-dependent. J Biol Chem 275: 7918–7924. - PubMed
-
- Alvarez A, Toro R, Caceres A, Maccioni RB (1999) Inhibition of tau phosphorylating protein kinase Cdk5 prevents β-amyloid-induced cell death. FEBS Lett 459: 421–426. - PubMed
-
- Anderson AJ, Cummings BJ, Cotman CW (1994) Increased immunoreactivity for Jun- and Fos-related proteins in Alzheimer's disease. Exp Neurol 125: 286–295. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
