Isoprenoid biosynthesis in the diatoms Rhizosolenia setigera (Brightwell) and Haslea ostrearia (Simonsen)
- PMID: 15070732
- PMCID: PMC384761
- DOI: 10.1073/pnas.0400902101
Isoprenoid biosynthesis in the diatoms Rhizosolenia setigera (Brightwell) and Haslea ostrearia (Simonsen)
Abstract
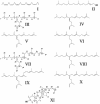
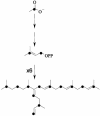
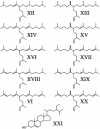
Isoprenoid biosynthesis in the widespread diatomaceous algae, Rhizosolenia setigera (Brightwell) and Haslea ostrearia (Simonsen), results not only in the production of diterpenoids, triterpenoids, and sterols but, unusually for diatoms, also in the production of sesterterpenoids. By using 13C and 2H isotopic labeling techniques followed by NMR and mass spectrometry, specific inhibition of mevalonate (MVA) and methylerythritol (MEP) pathways, and comparison with the natural 13C/12C isotope ratios of the lipids, the different biosynthetic pathways of the sesterterpenes and other isoprenoids have now been determined. Surprisingly, whereas the sesterterpenes (Delta(7(20))-haslenes) in R. setigera were made by the MVA pathway, as were the related triterpenoid rhizenes and desmosterol, in H. ostrearia the structurally similar Delta(6(17))-haslenes and the major sterol, 24-ethylcholest-5-en-3beta-ol, were instead biosynthesized by the MEP route. Phytol was biosynthesized in both diatoms by the MEP route. Subfractionation of R. setigera cells revealed that although phytol was located in the chloroplasts, the haslenes, rhizenes, and sterols were present in the cytoplasm. The observations described here for R. setigera and H. ostrearia show that terpenoid biosynthesis in diatoms is species-dependent and cannot simply be grouped according to structural type. Triterpenes appear to be made by the MVA route as in higher plants, whereas sesterterpenes and sterols can be made by either the MVA or MEP routes. In neither organism were the isoprenoids biosynthesized by leucine metabolism. Sesterterpene and triterpene biosynthesis in diatoms has not been investigated previously.
Figures


References
-
- Rohmer, M. (1999) Nat. Prod. Rep. 16, 565–574. - PubMed
-
- Ginger, M. L, Chance, M. L., Sadler, I. H. & Goad L. J. (2001) J. Biol. Chem. 276, 11674–11682. - PubMed
-
- Eisenreich, W., Schwarz, M., Cartayrade, A., Arigoni, D., Zenk, M. H. & Bacher, A. (1998) Chem. Biol. 5, 221–233. - PubMed
-
- Lichtenthaler, H. K. (1999) Annu. Rev. Plant Physiol. Plant Mol. Biol. 50, 47–65. - PubMed
-
- Bach, T. J. & Lichtenthaler, H. K. (1982) Z. Naturforsch. (C) 37, 46–50. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

