Real-time evaluation of myosin light chain kinase activation in smooth muscle tissues from a transgenic calmodulin-biosensor mouse
- PMID: 15071183
- PMCID: PMC395960
- DOI: 10.1073/pnas.0308742101
Real-time evaluation of myosin light chain kinase activation in smooth muscle tissues from a transgenic calmodulin-biosensor mouse
Abstract
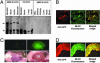
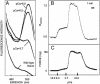
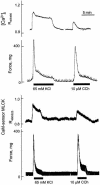
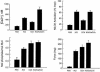
Ca(2+)/calmodulin (CaM)-dependent phosphorylation of myosin regulatory light chain (RLC) by myosin light chain kinase (MLCK) initiates smooth muscle contraction and regulates actomyosin-based cytoskeletal functions in nonmuscle cells. The net extent of RLC phosphorylation is controlled by MLCK activity relative to myosin light chain phosphatase activity. We have constructed a CaM-sensor MLCK where Ca(2+)-dependent CaM binding increases the catalytic activity of the kinase domain, whereas coincident binding to the biosensor domain decreases fluorescence resonance energy transfer between two fluorescent proteins. We have created transgenic mice expressing this construct specifically in smooth muscle cells to perform real-time evaluations of the relationship between smooth muscle contractility and MLCK activation in intact tissues and organs. Measurements in intact bladder smooth muscle demonstrate that MLCK activation increases rapidly during KCl-induced contractions but is not maximal, consistent with a limiting amount of cellular CaM. Carbachol treatment produces the same amount of force development and RLC phosphorylation, with much smaller increases in [Ca(2+)](i) and MLCK activation. A Rho kinase inhibitor suppresses RLC phosphorylation and force but not MLCK activation in carbachol-treated tissues. These observations are consistent with a model in which the magnitude of an agonist-mediated smooth muscle contraction depends on a rapid but limited Ca(2+)/CaM-dependent activation of MLCK and Rho kinase-mediated inhibition of myosin light chain phosphatase activity. These studies demonstrate the feasibility of producing transgenic biosensor mice for investigations of signaling processes in intact systems.
Figures

References
-
- Kamm, K. E. & Stull, J. T. (2001) J. Biol. Chem. 276, 4527-4530. - PubMed
-
- Somlyo, A. P. & Somlyo, A. V. (2003) Physiol. Rev. 83, 1325-1358. - PubMed
-
- Tansey, M. G., Luby-Phelps, K., Kamm, K. E. & Stull, J. T. (1994) J. Biol. Chem. 269, 9912-9920. - PubMed
-
- Tran, Q. K., Black, D. J. & Persechini, A. (2003) J. Biol. Chem. 278, 24247-24250. - PubMed
-
- Johnson, J. D., Snyder, C., Walsh, M. & Flynn, M. (1996) J. Biol. Chem. 271, 761-767. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

