Identification of cryptic MHC I-restricted epitopes encoded by HIV-1 alternative reading frames
- PMID: 15078897
- PMCID: PMC2211898
- DOI: 10.1084/jem.20031869
Identification of cryptic MHC I-restricted epitopes encoded by HIV-1 alternative reading frames
Abstract
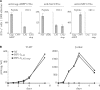
Human immunodeficiency virus (HIV) 1 major histocompatibility complex (MHC) I-restricted epitopes are widely believed to be derived from viral proteins encoded by primary open reading frames. However, the HIV-1 genome contains alternative reading frames (ARFs) potentially encoding small polypeptides. We have identified a panel of epitopes encoded by ARFs within the gag, pol, and env genes. The corresponding epitopic peptides were immunogenic in mice humanized for MHC-I molecules. In addition, cytotoxic T lymphocytes recognizing these epitopes were found in HIV-infected patients. These results reveal the existence of atypical mechanisms of HIV-1 epitope generation. They indicate that the repertoire of epitopes recognized by the cellular anti-HIV-1 immune response is broader than initially thought. This should be taken into account when designing vaccine strategies aimed at activating these responses.
Figures





References
-
- Schmitz, J.E., M.J. Kuroda, S. Santra, V.G. Sasseville, M.A. Simon, M.A. Lifton, P. Racz, K. Tenner-Racz, M. Dalesandro, B.J. Scallon, et al. 1999. Control of viremia in simian immunodeficiency virus infection by CD8+ lymphocytes. Science. 283:857–860. - PubMed
-
- Barouch, D.H., J. Kunstman, M.J. Kuroda, J.E. Schmitz, S. Santra, F.W. Peyerl, G.R. Krivulka, K. Beaudry, M.A. Lifton, D.A. Gorgone, et al. 2002. Eventual AIDS vaccine failure in a rhesus monkey by viral escape from cytotoxic T lymphocytes. Nature. 415:335–339. - PubMed
-
- Allen, T.M., D.H. O'Connor, P. Jing, J.L. Dzuris, B.R. Mothe, T.U. Vogel, E. Dunphy, M.E. Liebl, C. Emerson, N. Wilson, et al. 2000. Tat-specific cytotoxic T lymphocytes select for SIV escape variants during resolution of primary viraemia. Nature. 407:386–390. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

