Lipid rafts and integrin activation regulate oligodendrocyte survival
- PMID: 15084663
- PMCID: PMC6729358
- DOI: 10.1523/JNEUROSCI.5725-03.2004
Lipid rafts and integrin activation regulate oligodendrocyte survival
Abstract
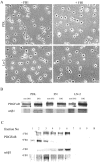
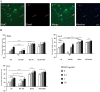
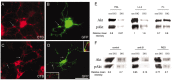
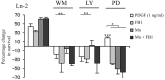
Newly formed oligodendrocytes in the CNS derive survival cues from their target axons. These cues are provided in part by laminins expressed on the axon, which are recognized by alpha6beta1 integrin on the oligdendrocyte and amplify platelet-derived growth factor (PDGF) signaling through the phosphatidylinositol 3'-kinase (PI3K) pathway. The alpha6beta1 integrin is localized in oligodendrocyte lipid rafts. We show here using the sphingolipid synthesis inhibitor fumonisin-B1 to deplete rafts that this localization is important for normal survival signaling, because depletion increases oligodendrocyte apoptosis and inhibits PI3K signaling. We have shown previously that PDGF-mediated integrin activation is an important component of oligodendrocyte proliferation signaling, and here we present evidence that a similar mechanism operates in survival signaling. Integrin activation using manganese increases raft localization and rescues the effects of both raft depletion and PDGF removal on survival and PI3K signaling. Together, these results point to an essential role for rafts in oligodendrocyte survival signaling on the basis of the provision of a favorable environment for growth factor-mediated integrin activation.
Figures


References
-
- Baron W, Decker L, Colognato H, ffrench-Constant C (2003) Regulation of integrin growth factor interactions in oligodendrocytes by lipid raft microdomains. Curr Biol 13: 151–155. - PubMed
-
- Barres BA, Raff MC (1994) Control of oligodendrocyte number in the developing rat optic nerve. Neuron 12: 935–942. - PubMed
-
- Barres BA, Hart IK, Coles HS, Burne JF, Voyvodic JT, Richardson WD, Raff MC (1992) Cell death and control of cell survival in the oligodendrocyte lineage. Cell 70: 31–46. - PubMed
-
- Bazzoni G, Hemler ME (1998) Are changes in integrin affinity and conformation overemphasized? Trends Biochem Sci 23: 30–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
