Aminoacylation properties of pathology-related human mitochondrial tRNA(Lys) variants
- PMID: 15100439
- PMCID: PMC1370574
- DOI: 10.1261/rna.5267604
Aminoacylation properties of pathology-related human mitochondrial tRNA(Lys) variants
Abstract
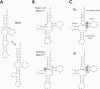
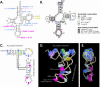
In vitro transcription has proven to be a successful tool for preparation of functional RNAs, especially in the tRNA field, in which, despite the absence of post-transcriptional modifications, transcripts are correctly folded and functionally active. Human mitochondrial (mt) tRNA(Lys) deviates from this principle and folds into various inactive conformations, due to the absence of the post-transcriptional modification m(1)A9 which hinders base-pairing with U64 in the native tRNA. Unavailability of a functional transcript is a serious drawback for structure/function investigations as well as in deciphering the molecular mechanisms by which point mutations in the mt tRNA(Lys) gene cause severe human disorders. Here, we show that an engineered in vitro transcribed "pseudo-WT" tRNA(Lys) variant is efficiently recognized by lysyl-tRNA synthetase and can substitute for the WT tRNA as a valuable reference molecule. This has been exploited in a systematic analysis of the effects on aminoacylation of nine pathology-related mutations described so far. The sole mutation located in a loop of the tRNA secondary structure, A8344G, does not affect aminoacylation efficiency. Out of eight mutations located in helical domains converting canonical Watson-Crick pairs into G-U pairs or C.A mismatches, six have no effect on aminoacylation (A8296G, U8316C, G8342A, U8356C, U8362G, G8363A), and two lead to drastic decreases (5000- to 7000-fold) in lysylation efficiencies (G8313A and G8328A). This screening, allowing for analysis of the primary impact level of all mutations affecting one tRNA under comparable conditions, indicates distinct molecular origins for different disorders.
Figures
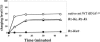



Similar articles
-
Comparative analysis of the pathogenic mechanisms associated with the G8363A and A8296G mutations in the mitochondrial tRNA(Lys) gene.Biochem J. 2005 May 1;387(Pt 3):773-8. doi: 10.1042/BJ20040949. Biochem J. 2005. PMID: 15554876 Free PMC article.
-
Wobble modification defect in tRNA disturbs codon-anticodon interaction in a mitochondrial disease.EMBO J. 2001 Sep 3;20(17):4794-802. doi: 10.1093/emboj/20.17.4794. EMBO J. 2001. PMID: 11532943 Free PMC article.
-
A double mutation (A8296G and G8363A) in the mitochondrial DNA tRNA (Lys) gene associated with myoclonus epilepsy with ragged-red fibers.Neurology. 1999 Jan 15;52(2):377-82. doi: 10.1212/wnl.52.2.377. Neurology. 1999. PMID: 9932960
-
Mitochondrial DNA G8363A mutation in the tRNA Lys gene: clinical, biochemical and pathological study.J Neurol Sci. 2009 Jun 15;281(1-2):85-92. doi: 10.1016/j.jns.2009.01.025. Epub 2009 Mar 10. J Neurol Sci. 2009. PMID: 19278689 Review.
-
Human mitochondrial diseases associated with tRNA wobble modification deficiency.RNA Biol. 2005 Apr;2(2):41-4. doi: 10.4161/rna.2.2.1610. Epub 2005 Apr 14. RNA Biol. 2005. PMID: 17132941 Review.
Cited by
-
Multifaceted roles of t6A biogenesis in efficiency and fidelity of mitochondrial gene expression.Nucleic Acids Res. 2024 Apr 12;52(6):3213-3233. doi: 10.1093/nar/gkae013. Nucleic Acids Res. 2024. PMID: 38227555 Free PMC article.
-
Molecular basis for human mitochondrial tRNA m3C modification by alternatively spliced METTL8.Nucleic Acids Res. 2022 Apr 22;50(7):4012-4028. doi: 10.1093/nar/gkac184. Nucleic Acids Res. 2022. PMID: 35357504 Free PMC article.
-
The concept of RNA-assisted protein folding: the role of tRNA.Theor Biol Med Model. 2012 Apr 2;9:10. doi: 10.1186/1742-4682-9-10. Theor Biol Med Model. 2012. PMID: 22462735 Free PMC article.
-
When a common biological role does not imply common disease outcomes: Disparate pathology linked to human mitochondrial aminoacyl-tRNA synthetases.J Biol Chem. 2019 Apr 5;294(14):5309-5320. doi: 10.1074/jbc.REV118.002953. Epub 2019 Jan 15. J Biol Chem. 2019. PMID: 30647134 Free PMC article. Review.
-
tRNA Metabolism and Neurodevelopmental Disorders.Annu Rev Genomics Hum Genet. 2019 Aug 31;20:359-387. doi: 10.1146/annurev-genom-083118-015334. Epub 2019 May 13. Annu Rev Genomics Hum Genet. 2019. PMID: 31082281 Free PMC article. Review.
References
-
- Agris, P.F. 1996. The importance of being modified: Roles of modified nucleosides and Mg2+ in RNA structure and function. Prog. Nucleic Acid Res. Mol. Biol. 53: 79–129. - PubMed
-
- Allen, J.F. and Raven, J.A. 1996. Free-radical-induced mutation vs redox regulation: Costs and benefits of genes in organelles. J. Mol. Evol. 42: 482–492. - PubMed
-
- Anderson, S., Bankier, A.T., Barrel, B.G., de Bruijn, M.H.L., Coulson, A.R., Drouin, J., Eperon, J.C., Nierlich, D.P., Roe, B.A., Sanger, F., et al. 1981. Sequence and organization of the human mitochondrial genome. Nature 290: 457–465. - PubMed
-
- Becker, H., Giegé, R., and Kern, D. 1996. Identity of prokaryotic and eukaryotic tRNAAsp for aminoacylation by aspartyl-tRNA synthetase from Thermus thermophilus. Biochemistry 35: 7447–7458. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
