Microsample preparation by dielectrophoresis: isolation of malaria
- PMID: 15100837
- PMCID: PMC2726252
- DOI: 10.1039/b110990c
Microsample preparation by dielectrophoresis: isolation of malaria
Abstract
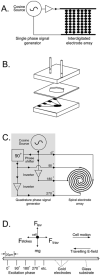
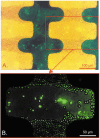
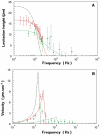
An important enabling factor for realising integrated micro fluidic analysis instruments for medical diagnostics purposes is front-end sample preparation. Dielectrophoresis is a method that offers great potential for cell discrimination and isolation for sample processing, and here we have applied it to the problem of isolating malaria-infected cells from blood. During development of the malarial pathogen, Plasmodium falciparum, increases occur in the ionic permeability of the plasma membrane of infected erythrocytes. When challenged by suspension in a low conductivity medium, infected cells lose internal ions while uninfected cells retain them. The resultant dielectric differences between infected and uninfected cells were exploited by dielectrophoretic manipulation in spatially inhomogeneous, travelling electrical fields produced by two types of microelectrode arrays. Parasitised cells of ring form or later stage from cultures and clinical specimens were isolated by steric dielectric field-flow-fractionation, focused at the centre of a spiral electrode array, and identified and counted. The dielectrophoretic methods require only a few micro litres of blood, and should be applicable to the production of small, low-cost automated devices for assessing parasite concentrations with potential applicability to drug sensitivity studies and the diagnosis of malaria. By simple adjustment of the electrical field parameters, other cell subpopulations that characterise disease, such as residual cancer cells in blood, can be similarly isolated and analysed.
Figures


Similar articles
-
Dielectrophoretic sorting on a microfabricated flow cytometer: label free separation of Babesia bovis infected erythrocytes.Bioelectrochemistry. 2008 Aug;73(2):123-8. doi: 10.1016/j.bioelechem.2008.04.018. Epub 2008 Apr 18. Bioelectrochemistry. 2008. PMID: 18511353
-
Dielectrophoretic detection of changes in erythrocyte membranes following malarial infection.Biochim Biophys Acta. 1997 Jan 31;1323(2):240-52. doi: 10.1016/s0005-2736(96)00191-5. Biochim Biophys Acta. 1997. PMID: 9042346
-
Isolation of erythrocytes infected with viable early stages of Plasmodium falciparum by flow cytometry.Cytometry A. 2012 Dec;81(12):1048-54. doi: 10.1002/cyto.a.22226. Epub 2012 Nov 7. Cytometry A. 2012. PMID: 23136095
-
Electric field-induced effects on neuronal cell biology accompanying dielectrophoretic trapping.Adv Anat Embryol Cell Biol. 2003;173:III-IX, 1-77. doi: 10.1007/978-3-642-55469-8. Adv Anat Embryol Cell Biol. 2003. PMID: 12901336 Review.
-
Ion metabolism in malaria-infected erythrocytes.Blood Cells. 1990;16(2-3):437-49. Blood Cells. 1990. PMID: 2175223 Review.
Cited by
-
Electronic detection of dielectrophoretic forces exerted on particles flowing over interdigitated electrodes.Biomicrofluidics. 2012 Jun;6(2):24117-2411715. doi: 10.1063/1.4709387. Epub 2012 May 3. Biomicrofluidics. 2012. PMID: 22655025 Free PMC article.
-
Implementation of an Integrated Dielectrophoretic and Magnetophoretic Microfluidic Chip for CTC Isolation.Biosensors (Basel). 2022 Sep 14;12(9):757. doi: 10.3390/bios12090757. Biosensors (Basel). 2022. PMID: 36140142 Free PMC article.
-
Dielectrophoretic-field flow fractionation analysis of dielectric, density, and deformability characteristics of cells and particles.Anal Chem. 2009 Nov 1;81(21):8878-85. doi: 10.1021/ac901470z. Anal Chem. 2009. PMID: 19791772 Free PMC article.
-
Dielectrophoretic separation of mouse melanoma clones.Biomicrofluidics. 2010 Jun 16;4(2):021101. doi: 10.1063/1.3447702. Biomicrofluidics. 2010. PMID: 20697600 Free PMC article.
-
Dielectrophoretic separation of blood cells.Biomed Microdevices. 2022 Aug 25;24(3):30. doi: 10.1007/s10544-022-00623-1. Biomed Microdevices. 2022. PMID: 36006519 Free PMC article. Review.
References
-
- Gascoyne PRC, Huang Y, Pethig R, Vykoukal J, Becker FF. Meas. Sci. Technol. 1992;3:439.
-
- Huang Y, Hölzel R, Pethig R, Wang X-B. Phys. Med. Biol. 1992;37:1499. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

