Complement protein C3 binding to Mycobacterium tuberculosis is initiated by the classical pathway in human bronchoalveolar lavage fluid
- PMID: 15102764
- PMCID: PMC387845
- DOI: 10.1128/IAI.72.5.2564-2573.2004
Complement protein C3 binding to Mycobacterium tuberculosis is initiated by the classical pathway in human bronchoalveolar lavage fluid
Abstract
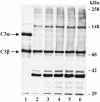
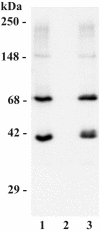
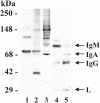
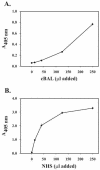
In high concentrations of fresh nonimmune human serum, Mycobacterium tuberculosis activates the alternative pathway of complement and binds C3 protein, resulting in enhanced phagocytosis by complement receptors on human alveolar macrophages. Yet in the lung, the alternative pathway of complement is relatively inactive compared to the classical pathway. To begin to determine whether C3 opsonophagocytosis of M. tuberculosis by alveolar macrophages can occur in the lung of the immunologically naive host, we characterized the binding of C3 to M. tuberculosis in different concentrations of fresh nonimmune human serum and concentrated human bronchoalveolar lavage fluid. Here we show that in human serum, C3 binding to M. tuberculosis is rapid, initiated by either the alternative pathway or the classical pathway, depending on the concentration of serum, and occurs by covalent linkages between the bacterial surface and the C3 cleavage products, C3b or C3bi. Human bronchoalveolar lavage fluid contains C3 protein and functional classical pathway activity that mediates the binding of C3 to the surface of M. tuberculosis. These studies provide evidence that when M. tuberculosis is first inhaled into the lungs of the human host, the bacterium is opsonized by C3 cleavage via classical pathway activation within the alveolus, providing a C3-dependent entry pathway into resident alveolar macrophages.
Figures





References
-
- Actor, J. K., E. Breij, R. A. Wetsel, H. Hoffmann, R. L. Hunter, Jr., and C. Jagannath. 2001. A role for complement C5 in organism containment and granulomatous response during murine tuberculosis. Scand. J. Immunol. 53:464-474. - PubMed
-
- Berger, M. 1990. Third component of human complement: C3. Methods Enzymol. 184:619-628. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

