Nitric oxide-mediated regulation of gamma interferon-induced bacteriostasis: inhibition and degradation of human indoleamine 2,3-dioxygenase
- PMID: 15102781
- PMCID: PMC387869
- DOI: 10.1128/IAI.72.5.2723-2730.2004
Nitric oxide-mediated regulation of gamma interferon-induced bacteriostasis: inhibition and degradation of human indoleamine 2,3-dioxygenase
Abstract
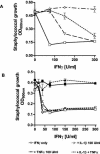
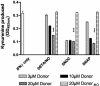
Tryptophan depletion resulting from indoleamine 2,3-dioxygenase (IDO) activity within the kynurenine pathway is one of the most prominent gamma interferon (IFN-gamma)-inducible antimicrobial effector mechanisms in human cells. On the other hand, nitric oxide (NO) produced by the inducible isoform of NO synthase (iNOS) serves a more immunoregulatory role in human cells and thereby interacts with tryptophan depletion in a number of ways. We investigated the effects of NO on IDO gene transcription, protein synthesis, and enzyme activity as well as on IDO-mediated bacteriostasis in the human epithelial cell line RT4. IFN-gamma-stimulated RT4 cells were able to inhibit the growth of Staphylococcus aureus in an IDO-mediated fashion, and this bacteriostatic effect was abolished by endogenously produced NO. These findings were supported by experiments which showed that IDO activity in extracts of IFN-gamma-stimulated cells is inhibited by the chemical NO donors diethylenetriamine diazeniumdiolate, S-nitroso-L-cysteine, and S-nitroso-N-acetyl-D,L-penicillamine. Furthermore, we found that both endogenous and exogenous NO strongly reduced the level of IDO protein content in RT4 cells. This effect was not due to a decrease in IDO gene transcription or mRNA stability. By using inhibitors of proteasomal proteolytic activity, we showed that NO production led to an accelerated degradation of IDO protein in the proteasome. This is the first report, to our knowledge, that demonstrates that the IDO is degraded by the proteasome and that NO has an effect on IDO protein stability.
Figures




References
-
- Alberati-Giani, D., P. Malherbe, P. Ricciardi-Castagnoli, C. Köhler, S. Denis-Donini, and A. M. Cesura. 1997. Differential regulation of indoleamine-2,3-dioxygenase expression by nitric oxide and inflammatory mediators in IFN-γ-activated murine macrophages and microglial cells. J. Immunol. 159:419-426. - PubMed
-
- Bebok, Z., K. Varga, J. K. Hicks, C. J. Venglarik, T. Kovacs, L. Chen, K. M. Hardiman, J. F. Collawn, E. J. Sorscher, and S. Matalon. 2002. Reactive oxygen nitrogen species decrease cystic fibrosis transmembrane conductance regulator expression and cAMP-mediated Cl− secretion in airway epithelia. J. Biol. Chem. 277:43041-43049. - PubMed
-
- Bingisser, R. M., T. A. Tilbrook, P. G. Holt, and U. R. Kees. 1998. Macrophage-derived nitric oxide regulates T cell activation via reversible disruption of the Jak3/STAT5 signaling pathway. J. Immunol. 160:5729-5734. - PubMed
-
- Bogdan, C., M. Röllinghoff, and A. Diefenbach. 2000. Reactive oxygen and reactive nitrogen intermediates in innate and specific immunity. Curr. Opin. Immunol. 12:64-76. - PubMed
-
- Byrne, G. I., L. K. Lehmann, J. G. Kirschbaum, E. C. Borden, C. M. Lee, and R. R. Brown. 1986. Induction of tryptophan degradation in vitro and in vivo: a g-Interferon-stimulated activity. J. Interferon Res. 6:389-396. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

