Fungus-elicited metabolites from plants as an enriched source for new leishmanicidal agents: antifungal phenyl-phenalenone phytoalexins from the banana plant (Musa acuminata) target mitochondria of Leishmania donovani promastigotes
- PMID: 15105102
- PMCID: PMC400542
- DOI: 10.1128/AAC.48.5.1534-1540.2004
Fungus-elicited metabolites from plants as an enriched source for new leishmanicidal agents: antifungal phenyl-phenalenone phytoalexins from the banana plant (Musa acuminata) target mitochondria of Leishmania donovani promastigotes
Abstract
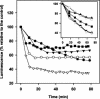
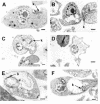
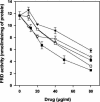
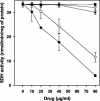
Two antifungal phenyl-phenalenone phytoalexins isolated from the banana plant (Musa acuminata) elicited with the fungus Fusarium oxysporum, together with a methoxy derivative of one of them and two epoxide precursors of their chemical synthesis, were tested for leishmanicidal activity on Leishmania donovani promastigotes and L. infantum amastigotes. Drugs inhibited proliferation of both forms of the parasite with a 50% lethal concentration range between 10.3 and 68.7 micro g/ml. Their lethal mechanism was found linked to the respiratory chain by a systematic approach, including electron microscopy, measurement of the oxygen consumption rate on digitonin-permeabilized promastigotes, and enzymatic assays on a mitochondrial enriched fraction. Whereas the whole set of compounds inhibited the activity of fumarate reductase in the mitochondrial fraction (50% effective concentration [EC(50)] between 33.3 and 78.8 micro g/ml) and on purified enzyme (EC(50) = 53.3 to 115 micro g/ml), inhibition for succinate dehydrogenase was only observed for the two phytoalexins with the highest leishmanicidal activity: anigorufone and its natural analogue 2-methoxy-9-phenyl-phenalen-1-one (EC(50) = 33.5 and 59.6 micro g/ml, respectively). These results provided a new structural motif, phenyl-phenalenone, as a new lead for leishmanicidal activity, and support the use of plant extracts enriched in antifungal phytoalexins, synthesized under fungal challenge, as a more rational and effective strategy to screen for new plant leishmanicidal drugs.
Figures

References
-
- Álvarez-Fortes, E., L. M. Ruiz-Pérez, F. Bouillaud, E. Rial, and L. Rivas. 1998. Expression and regulation of mitochondrial uncoupling protein 1 from brown adipose tissue in Leishmania major promastigotes. Mol. Biochem. Parasitol. 93:191-202. - PubMed
-
- Armson, A., S. W. Kamau, F. Grimm, J. A. Reynoldson, W. M. Best, L. M. MacDonald, and R. C. Thompson. 1999. A comparison of the effects of a benzimidazole and the dinitroanilines against Leishmania infantum. Acta Trop. 73:303-311. - PubMed
-
- Balaña-Fouce, R., R. M. Reguera, J. C. Cubría, and D. Ordóñez. 1998. The pharmacology of leishmaniasis. Gen. Pharmacol. 30:435-443. - PubMed
-
- Bermúdez, R., F. Dagger, J. A. D'Aquino, G. Benaim, and K. Dawidowicz. 1997. Characterization of mitochondrial electron-transfer in Leishmania mexicana. Mol. Biochem. Parasitol. 90:43-54. - PubMed
-
- Blum, J. J. 1993. Intermediary metabolism of Leishmania. Parasitol. Today 9:118-122. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

