Endothelial cells present antigens in vivo
- PMID: 15113397
- PMCID: PMC394319
- DOI: 10.1186/1471-2172-5-5
Endothelial cells present antigens in vivo
Abstract
Background: Immune recognition of vascular endothelial cells (EC) has been implicated in allograft rejection, protection against pathogens, and lymphocyte recruitment. However, EC pervade nearly all tissues and predominate in none, complicating any direct test of immune recognition. Here, we examined antigen presentation by EC in vivo by testing immune responses against E. coli beta-galactosidase (beta-gal) in two lines of transgenic mice that express beta-gal exclusively in their EC. TIE2-lacZ mice express beta-gal in all EC and VWF-lacZ mice express beta-gal in heart and brain microvascular EC.
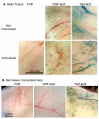
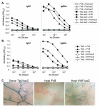
Results: Transgenic and congenic wild type FVB mice immunized with beta-gal expression vector DNA or beta-gal protein generated high titer, high affinity antisera containing comparable levels of antigen-specific IgG1 and IgG2a isotypes, suggesting equivalent activation of T helper cell subsets. The immunized transgenic mice remained healthy, their EC continued to express beta-gal, and their blood vessels showed no histological abnormalities. In response to beta-gal in vitro, CD4+ and CD8+ T cells from immunized transgenic and FVB mice proliferated, expressed CD25, and secreted IFN-gamma. Infection with recombinant vaccinia virus encoding beta-gal raised equivalent responses in transgenic and FVB mice. Hearts transplanted from transgenic mice into FVB mice continued to beat and the graft EC continued to express beta-gal. These results suggested immunological ignorance of the transgene encoded EC protein. However, skin transplanted from TIE2-lacZ onto FVB mice lost beta-gal+ EC and the hosts developed beta-gal-specific antisera, demonstrating activation of host immune effector mechanisms. In contrast, skin grafted from TIE2-lacZ onto VWF-lacZ mice retained beta-gal+ EC and no antisera developed, suggesting a tolerant host immune system.
Conclusion: Resting, beta-gal+ EC in transgenic mice tolerize specific lymphocytes that would otherwise respond against beta-gal expressed by EC within transplanted skin. We conclude that EC effectively present intracellular "self" proteins to the immune system. However, antigen presentation by EC does not delete or anergize a large population of specific lymphocytes that respond to the same protein following conventional immunization with protein or expression vector DNA. These results clearly demonstrate striking context sensitivity in the immune recognition of EC, a subtlety that must be better understood in order to treat immune diseases and complications involving the vasculature.
Figures




References
-
- Pober JS, Cotran RS. The role of endothelial cells in inflammation. Transplantation. 1990;50:537–544. - PubMed
-
- de Waal RM, Bogman MJ, Maass CN, Cornelissen LM, Tax WJ, Koene RA. Variable expression of Ia antigens on the vascular endothelium of mouse skin allografts. Nature. 1983;303:426–429. - PubMed
-
- Racusen LC, Solez K, Colvin RB, Bonsib SM, Castro MC, Cavallo T, Croker BP, Demetris AJ, Drachenberg CB, Fogo AB, Furness P, Gaber LW, Gibson IW, Glotz D, Goldberg JC, Grande J, Halloran PF, Hansen HE, Hartley B, Hayry PJ, Hill CM, Hoffman EO, Hunsicker LG, Lindblad AS, Yamaguchi Y, et al. The Banff 97 working classification of renal allograft pathology. Kidney Int. 1999;55:713–723. doi: 10.1046/j.1523-1755.1999.00299.x. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

