Degradative organelles containing mislocalized alpha-and beta-synuclein proliferate in presenilin-1 null neurons
- PMID: 15123735
- PMCID: PMC2172178
- DOI: 10.1083/jcb.200403061
Degradative organelles containing mislocalized alpha-and beta-synuclein proliferate in presenilin-1 null neurons
Abstract
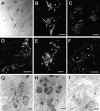
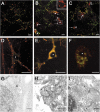
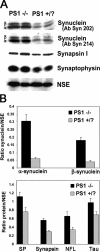
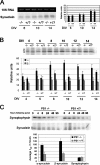
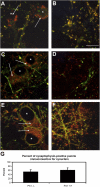
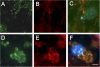
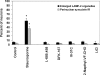
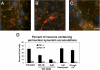
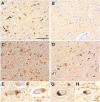
Presenilin-1 null mutation (PS1 -/-) in mice is associated with morphological alterations and defects in cleavage of transmembrane proteins. Here, we demonstrate that PS1 deficiency also leads to the formation of degradative vacuoles and to the aberrant translocation of presynaptic alpha- and beta-synuclein proteins to these organelles in the perikarya of primary neurons, concomitant with significant increases in the levels of both synucleins. Stimulation of autophagy in control neurons produced a similar mislocalization of synucleins as genetic ablation of PS1. These effects were not the result of the loss of PS1 gamma-secretase activity; however, dysregulation of calcium channels in PS1 -/- cells may be involved. Finally, colocalization of alpha-synuclein and degradative organelles was observed in brains from patients with the Lewy body variant of AD. Thus, aberrant accumulation of alpha- and beta-synuclein in degradative organelles are novel features of PS1 -/- neurons, and similar events may promote the formation of alpha-synuclein inclusions associated with neurodegenerative diseases.
Copyright the Rockefeller University Press
Figures

References
-
- Adamec, E., P.S. Mohan, A.M. Cataldo, A.N. Srinivasan, J.P. Vonsattel, and R.A. Nixon. 2000. Upregulation of the lysosomal system in experimental models of neuronal injury: Implications for Alzheimer's disease. Neuroscience. 100:663–675. - PubMed
-
- Anglade, P., S. Vyas, F. Javoy-Agid, M.T. Herrero, P.P. Michel, J. Marquez, A. Mouatt-Prigent, M. Ruberg, E.C. Hirsch, and Y. Agid. 1997. Apoptosis and autophagy in nigral neurons of patients with Parkinson's disease. Histol. Histopathol. 12:25–31. - PubMed
-
- Annaert, W.G., C. Esselens, V. Baert, C. Boeve, G. Snellings, P. Cupers, K. Craessaerts, and B. De Strooper. 2001. Interaction with telencephalin and the amyloid precursor protein predicts a ring structure for presenilins. Neuron. 32:579–589. - PubMed
-
- Bembi, B., M.S. Zambito, E. Sidransky, G. Ciana, M. Carrozzi, C. Martini, M. Giolis, M.G. Pittis, and L. Capus. 2003. Gaucher's disease with Parkinson's disease: Clinical and pathogical aspects. Neurology. 61:99–101. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

