Decreased sialylation of the acute phase protein alpha1-acid glycoprotein in feline infectious peritonitis (FIP)
- PMID: 15135988
- PMCID: PMC7120068
- DOI: 10.1016/j.vetimm.2004.02.003
Decreased sialylation of the acute phase protein alpha1-acid glycoprotein in feline infectious peritonitis (FIP)
Abstract
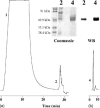
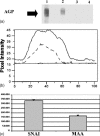
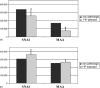
Feline infectious peritonitis (FIP) is an immune-mediated disease of domestic and exotic felides infected with feline coronavirus. FIP is characterized by the overexpression of an acute phase protein, the alpha1-acid glycoprotein (AGP). In humans, AGP is a heavily glycosylated protein that undergoes several modifications of its glycan moiety during acute and chronic inflammatory pathologies. We studied the changes in AGP glycosylation in the course of FIP. Specifically, we focussed our attention on the degree of sialylation, fucosylation and branching. This study presents a purification method for feline AGP (fAGP) from serum, using an ion exchange chromatography strategy. The glycosylation pattern was analyzed in detail by means of interaction of purified fAGP with specific lectins. In particular, Sambucus nigra agglutinin I and Maackia amurensis agglutinin lectins were used to detect sialic acid residues, Aleuria aurantia lectin was used to detect L-fucose residues and Concanavalin A was used to evaluate the branching degree. By this method we showed that fAGP did not present any L-fucose residues on its surface, and that its branching degree was very low, both in normal and in pathological conditions. In contrast, during FIP disease, fAGP underwent several modifications in the sialic acid content, including decreased expression of both alpha(2-6)-linked and alpha(2-3)-linked sialic acid (76 and 44%, respectively when compared to non-pathological feline AGP).
Copyright 2004 Elsevier B.V.
Figures

Similar articles
-
Glycan moiety modifications of feline alpha1-acid glycoprotein in retrovirus (FIV, FeLV) affected cats.Vet Immunol Immunopathol. 2005 Aug 15;107(1-2):17-26. doi: 10.1016/j.vetimm.2005.02.021. Vet Immunol Immunopathol. 2005. PMID: 15982476
-
Hyposialylated α1-acid glycoprotein inhibits phagocytosis of feline neutrophils.Res Vet Sci. 2013 Oct;95(2):465-71. doi: 10.1016/j.rvsc.2013.04.026. Epub 2013 May 31. Res Vet Sci. 2013. PMID: 23726663 Free PMC article.
-
Expression patterns in feline blood and tissues of α1-acid glycoprotein (AGP) and of an AGP-related protein (AGPrP).Comp Clin Path. 2003;12(3):140-146. doi: 10.1007/s00580-003-0489-8. Comp Clin Path. 2003. PMID: 32214964 Free PMC article.
-
Carbohydrate composition and immunomodulatory activity of different glycoforms of alpha1-acid glycoprotein.Glycoconj J. 1997 Aug;14(5):631-8. doi: 10.1023/a:1018544711767. Glycoconj J. 1997. PMID: 9298696 Review.
-
Feline infectious peritonitis: still an enigma?Vet Pathol. 2014 Mar;51(2):505-26. doi: 10.1177/0300985814522077. Vet Pathol. 2014. PMID: 24569616 Review.
Cited by
-
The paradox of feline coronavirus pathogenesis: a review.Adv Virol. 2011;2011:109849. doi: 10.1155/2011/109849. Epub 2011 Aug 21. Adv Virol. 2011. PMID: 22312333 Free PMC article.
-
Serum alpha1-acid glycoprotein (AGP) concentration in non-symptomatic cats with feline coronavirus (FCoV) infection.J Feline Med Surg. 2007 Aug;9(4):271-7. doi: 10.1016/j.jfms.2007.01.002. Epub 2007 Mar 6. J Feline Med Surg. 2007. PMID: 17344083 Free PMC article.
-
Feline infectious peritonitis.Vet Clin North Am Small Anim Pract. 2005 Jan;35(1):39-79, vi. doi: 10.1016/j.cvsm.2004.10.011. Vet Clin North Am Small Anim Pract. 2005. PMID: 15627627 Free PMC article. Review.
-
Total sialic acid: an acute phase reactant in cats with a possible role in feline coronavirus infection.Can J Vet Res. 2009 Apr;73(2):144-50. Can J Vet Res. 2009. PMID: 19436584 Free PMC article.
-
Feline Infectious Peritonitis: European Advisory Board on Cat Diseases Guidelines.Viruses. 2023 Aug 31;15(9):1847. doi: 10.3390/v15091847. Viruses. 2023. PMID: 37766254 Free PMC article. Review.
References
-
- Bories P.N, Feger J, Benbernou N, Rouzeau J.D, Agneray J, Durand G. Prevalence of tri- and tetraantennary glycans of human alpha 1-acid glycoprotein in release of macrophage inhibitor of interleukin-1 activity. Inflammation. 1990;14:315–323. - PubMed
-
- Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities or protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. - PubMed
-
- Brinkman-van der Linden E.C, van Ommen E.C, van Dijk W. Glycosylation of alpha 1-acid glycoprotein in septic shock: changes in degree of branching and in expression of sialyl Lewis(x) groups. Glycoconjugate J. 1996;13:27–31. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

