Study of humoral immunity to commensal oral bacteria in human infants demonstrates the presence of secretory immunoglobulin A antibodies reactive with Actinomyces naeslundii genospecies 1 and 2 ribotypes
- PMID: 15138172
- PMCID: PMC404577
- DOI: 10.1128/CDLI.11.3.473-482.2004
Study of humoral immunity to commensal oral bacteria in human infants demonstrates the presence of secretory immunoglobulin A antibodies reactive with Actinomyces naeslundii genospecies 1 and 2 ribotypes
Abstract
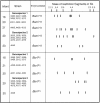
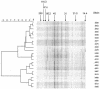
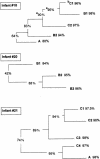
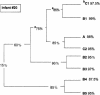
The mouths of three human infants were examined from birth to age 2 years to detect colonization of Actinomyces naeslundii genospecies 1 and 2. These bacteria did not colonize until after tooth eruption. The diversity of posteruption isolates was determined by ribotyping. Using immunoblotting and enzyme-linked immunosorbent assay, we determined the reactivity of secretory immunoglobulin A (SIgA) antibodies in saliva samples collected from each infant before and after colonization against cell wall proteins from their own A. naeslundii strains and carbohydrates from standard A. naeslundii genospecies 1 and 2 strains. A. naeslundii genospecies 1 and 2 carbohydrate-reactive SIgA antibodies were not detected in any saliva sample. However, SIgA antibodies reactive with cell wall proteins were present in saliva before these bacteria colonized the mouth. These antibodies could be almost completely removed by absorption with A. odontolyticus, a species known to colonize the human mouth shortly after birth. However, after colonization by A. naeslundii genospecies 1 and 2, specific antibodies were induced that could not be removed by absorption with A. odontolyticus. Cluster analysis of the patterns of reactivity of postcolonization salivary antibodies from each infant with antigens from their own strains showed that not only could these antibodies discriminate among strains but antibodies in saliva samples collected at different times showed different reactivity patterns. Overall, these data suggest that, although much of the salivary SIgA antibodies reactive with A. naeslundii genospecies 1 and 2 are directed against genus-specific or more broadly cross-reactive antigens, species, genospecies, and possibly strain-specific antibodies are induced in response to colonization.
Figures

Similar articles
-
Humoral immunity to commensal oral bacteria in human infants: salivary antibodies reactive with Actinomyces naeslundii genospecies 1 and 2 during colonization.Infect Immun. 1998 Sep;66(9):4283-9. doi: 10.1128/IAI.66.9.4283-4289.1998. Infect Immun. 1998. PMID: 9712779 Free PMC article.
-
Humoral immunity to commensal oral bacteria in human infants: evidence that Streptococcus mitis biovar 1 colonization induces strain-specific salivary immunoglobulin A antibodies.ISME J. 2008 Jul;2(7):728-38. doi: 10.1038/ismej.2008.26. Epub 2008 Apr 17. ISME J. 2008. PMID: 18421259
-
Salivary IgA antibody responses to Streptococcus mitis and Streptococcus mutans in preterm and fullterm newborn children.Arch Oral Biol. 2012 Jun;57(6):647-53. doi: 10.1016/j.archoralbio.2011.11.011. Epub 2011 Dec 12. Arch Oral Biol. 2012. PMID: 22169809
-
[Cellular and humoral mechanism of the antimicrobial protection of the oral cavity].Stomatologiia (Mosk). 1977 Sep-Oct;56(5):91-8. Stomatologiia (Mosk). 1977. PMID: 333665 Review. Russian. No abstract available.
-
Oral microbial ecology and the role of salivary immunoglobulin A.Microbiol Mol Biol Rev. 1998 Mar;62(1):71-109. doi: 10.1128/MMBR.62.1.71-109.1998. Microbiol Mol Biol Rev. 1998. PMID: 9529888 Free PMC article. Review.
Cited by
-
Assessment of Age-Related Changes of Salivary Immunoglobulin A Levels among Healthy Individuals.J Pharm Bioallied Sci. 2021 Jun;13(Suppl 1):S461-S464. doi: 10.4103/jpbs.JPBS_605_20. Epub 2021 Jun 5. J Pharm Bioallied Sci. 2021. PMID: 34447134 Free PMC article.
-
The Application of Magnetic Bead Selection to Investigate Interactions between the Oral Microbiota and Salivary Immunoglobulins.PLoS One. 2016 Aug 2;11(8):e0158288. doi: 10.1371/journal.pone.0158288. eCollection 2016. PLoS One. 2016. PMID: 27483159 Free PMC article.
References
-
- Bolken, T. C., C. A. Franze, K. F. Jones, R. H. Bell, R. M. Swanson, D. S. King, V. A. Fischetti, and D. E. Hruby. 2002. Analysis of factors affecting surface expression and immunogenicity of recombinant proteins expressed by gram-positive commensal vectors. Infect. Immun. 70:2487-2491. - PMC - PubMed
-
- Bowden, G., J. Johnson, and C. Schachtele. 1993. Characterization of Actinomyces with genomic DNA fingerprints and rRNA gene probes. J. Dent. Res. 72:1171-1179. - PubMed
-
- Bowden, G. H., N. Nolette, H. Ryding, and B. M. Cleghorn. 1999. The diversity and distribution of the predominant ribotypes of Actinomyces naeslundii genospecies 1 and 2 in samples from enamel and from healthy and carious root surfaces of teeth. J. Dent. Res. 78:1800-1809. - PubMed
-
- Bowden, G. H. W. 1998. Actinomyces, p. 445-462. In A. Balows and B. I. Duerden (ed.), Topley and Wilson's microbiology and microbial infections, 9th ed. Arnold, London, England.
-
- Bowden, G. H. W., J. M. Hardie, and E. D. Fillery. 1976. Antigens from Actinomyces species and their value in identification. J. Dent. Res. 55A:192-204. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

