The Abl-related gene (Arg) requires its F-actin-microtubule cross-linking activity to regulate lamellipodial dynamics during fibroblast adhesion
- PMID: 15138293
- PMCID: PMC2172189
- DOI: 10.1083/jcb.200308055
The Abl-related gene (Arg) requires its F-actin-microtubule cross-linking activity to regulate lamellipodial dynamics during fibroblast adhesion
Abstract
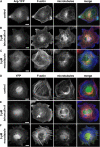
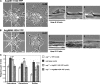
Microtubules (MTs) help establish and maintain cell polarity by promoting actin-dependent membrane protrusion at the leading edge of the cell, but the molecular mechanisms that mediate cross-talk between actin and MTs during this process are unclear. We demonstrate that the Abl-related gene (Arg) nonreceptor tyrosine kinase is required for dynamic lamellipodial protrusions after adhesion to fibronectin. arg-/- fibroblasts exhibit reduced lamellipodial dynamics as compared with wild-type fibroblasts, and this defect can be rescued by reexpression of an Arg-yellow fluorescent protein fusion. We show that Arg can bind MTs with high affinity and cross-link filamentous actin (F-actin) bundles and MTs in vitro. MTs concentrate and insert into Arg-induced F-actin-rich cell protrusions. Arg requires both its F-actin-binding domains and its MT-binding domain to rescue the defects in lamellipodial dynamics of arg-/- fibroblasts. These findings demonstrate that Arg can mediate physical contact between F-actin and MTs at the cell periphery and that this cross-linking activity is required for Arg to regulate lamellipodial dynamics in fibroblasts.
Copyright the Rockefeller University Press
Figures


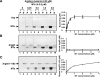
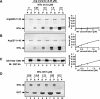
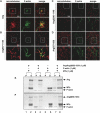
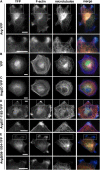
References
-
- Bashaw, G.J., T. Kidd, D. Murray, T. Pawson, and C.S. Goodman. 2000. Repulsive axon guidance: Abelson and Enabled play opposing roles downstream of the roundabout receptor. Cell. 101:703–715. - PubMed
-
- Bershadsky, A.D., E.A. Vaisberg, and J.M. Vasiliev. 1991. Pseudopodial activity at the active edge of migrating fibroblast is decreased after drug-induced microtubule depolymerization. Cell Motil. Cytoskeleton. 19:152–158. - PubMed
-
- Blanchoin, L., and T.D. Pollard. 1998. Interaction of actin monomers with Acanthamoeba actophorin (ADF/cofilin) and profilin. J. Biol. Chem. 273:25106–25111. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

