Fatal disseminated mouse adenovirus type 1 infection in mice lacking B cells or Bruton's tyrosine kinase
- PMID: 15140955
- PMCID: PMC415807
- DOI: 10.1128/JVI.78.11.5584-5590.2004
Fatal disseminated mouse adenovirus type 1 infection in mice lacking B cells or Bruton's tyrosine kinase
Abstract
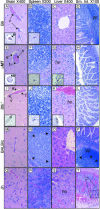
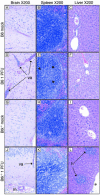
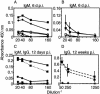
Mouse adenovirus type 1 (MAV-1) infection of B-cell-deficient and Bruton's tyrosine kinase (Btk)-deficient mice resulted in fatal disseminated disease resembling human adenovirus infections in immunocompromised patients. Mice lacking B cells or Btk were highly susceptible to acute MAV-1 infection, in contrast to controls and mice lacking T cells. To our knowledge, this is the first demonstration that mice with an X-linked immunodeficiency phenotype (Btk deficient) are susceptible to virus-induced disease. Mice lacking B cells or Btk on a C57BL/6 background succumbed with encephalomyelitis, hepatitis, and lymphoid necrosis. Mice lacking B cells on a BALB/c background succumbed with enteritis and hepatitis. Survival of acute MAV-1 infection correlated with early T-cell-independent neutralizing antibody and T-cell-independent antiviral immunoglobulin M. Treatment of MAV-1-infected Btk(-/-) mice 4 to 9 days postinfection with antiserum harvested 6 to 9 days postinfection from MAV-1-infected Btk(+/+) mice was therapeutic. Our findings implicate a critical role for B-cell function in preventing disseminated MAV-1 infection, particularly production of early T-cell-independent antiviral immunoglobulin M.
Figures





Similar articles
-
T cells cause acute immunopathology and are required for long-term survival in mouse adenovirus type 1-induced encephalomyelitis.J Virol. 2003 Sep;77(18):10060-70. doi: 10.1128/jvi.77.18.10060-10070.2003. J Virol. 2003. PMID: 12941916 Free PMC article.
-
Recovery of humoral immunity is critical for successful antiviral therapy in disseminated mouse adenovirus type 1 infection.Antimicrob Agents Chemother. 2008 Apr;52(4):1462-71. doi: 10.1128/AAC.01311-07. Epub 2008 Feb 11. Antimicrob Agents Chemother. 2008. PMID: 18268085 Free PMC article.
-
The Bruton's tyrosine kinase gene is expressed throughout B cell differentiation, from early precursor B cell stages preceding immunoglobulin gene rearrangement up to mature B cell stages.Eur J Immunol. 1993 Dec;23(12):3109-14. doi: 10.1002/eji.1830231210. Eur J Immunol. 1993. PMID: 8258324
-
The role of Bruton's tyrosine kinase in B-cell development and function: a genetic perspective.Immunol Rev. 2000 Jun;175:120-7. Immunol Rev. 2000. PMID: 10933597 Review.
-
Bruton's tyrosine kinase (Btk)-the critical tyrosine kinase in LPS signalling?Immunol Lett. 2004 Mar 29;92(1-2):15-22. doi: 10.1016/j.imlet.2003.11.017. Immunol Lett. 2004. PMID: 15081522 Review.
Cited by
-
Defensins Potentiate a Neutralizing Antibody Response to Enteric Viral Infection.PLoS Pathog. 2016 Mar 2;12(3):e1005474. doi: 10.1371/journal.ppat.1005474. eCollection 2016 Mar. PLoS Pathog. 2016. PMID: 26933888 Free PMC article.
-
Susceptibility to acute mouse adenovirus type 1 respiratory infection and establishment of protective immunity in neonatal mice.J Virol. 2012 Apr;86(8):4194-203. doi: 10.1128/JVI.06967-11. Epub 2012 Feb 15. J Virol. 2012. PMID: 22345470 Free PMC article.
-
Immunological pathogenesis and treatment progress of adenovirus pneumonia in children.Ital J Pediatr. 2025 Jan 9;51(1):4. doi: 10.1186/s13052-024-01836-1. Ital J Pediatr. 2025. PMID: 39789604 Free PMC article. Review.
-
Role of mouse adenovirus type 1 E4orf6-induced degradation of protein kinase R in pathogenesis.J Virol. 2025 Feb 25;99(2):e0154524. doi: 10.1128/jvi.01545-24. Epub 2024 Dec 31. J Virol. 2025. PMID: 39745442 Free PMC article.
-
Mouse adenovirus type 1 infection in SCID mice: an experimental model for antiviral therapy of systemic adenovirus infections.Antimicrob Agents Chemother. 2005 Nov;49(11):4689-99. doi: 10.1128/AAC.49.11.4689-4699.2005. Antimicrob Agents Chemother. 2005. PMID: 16251313 Free PMC article.
References
-
- Balkow, S., A. Kersten, T. T. Tran, T. Stehle, P. Grosse, C. Museteanu, O. Utermohlen, H. Pircher, F. von Weizsacker, R. Wallich, A. Mullbacher, and M. M. Simon. 2001. Concerted action of the FasL/Fas and perforin/granzyme A and B pathways is mandatory for the development of early viral hepatitis but not for recovery from viral infection. J. Virol. 75:8781-8791. - PMC - PubMed
-
- Baumgarth, N. 2000. A two-phase model of B-cell activation. Immunol. Rev. 176:171-180. - PubMed
-
- Berke, Z., H. Mellin, S. Heidari, T. Wen, A. Berglof, G. Klein, and T. Dalianis. 1998. Adult X-linked immunodeficiency (XID) mice, IGM-/- single knockout and IGM-/-CD8-/- double knockout mice do not clear polyomavirus infection. In Vivo 12:143-148. - PubMed
-
- Blanke, C., C. Clark, E. R. Broun, G. Tricot, I. Cunningham, K. Cornetta, A. Hedderman, and R. Hromas. 1995. Evolving pathogens in allogeneic bone marrow transplantation: increased fatal adenoviral infections. Am. J. Med. 99:326-328. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

