Mechanosensory S-neurons rather than AH-neurons appear to generate a rhythmic motor pattern in guinea-pig distal colon
- PMID: 15146052
- PMCID: PMC1664963
- DOI: 10.1113/jphysiol.2004.063586
Mechanosensory S-neurons rather than AH-neurons appear to generate a rhythmic motor pattern in guinea-pig distal colon
Abstract
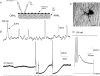
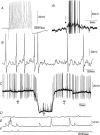
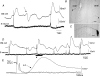
Simultaneous intracellular recordings were made from myenteric neurons and circular muscle (CM) cells in isolated, stretched segments of guinea-pig distal colon. We have shown previously that maintained stretch generates a repetitive and coordinated discharge of ascending excitatory and descending inhibitory neuronal reflex pathways in the distal colon. In the presence of nifedipine (1-2 microm) to paralyse the muscle, simultaneous recordings were made from 25 pairs of AH (after-hyperpolarization)-neurons and CM cells separated by 100-500 microm. In all 25 AH-neurons, proximal process potentials (PPPs) were never recorded, even though at the same time, all recordings from neighbouring CM cells showed an ongoing discharge of inhibitory junction potentials (IJPs) anally, or excitatory junction potentials (EJPs) orally. In fact, 24 of 25 AH-neurons were totally silent, while in one AH-cell, some spontaneous fast excitatory postsynaptic potentials (FEPSPs) were recorded. All 10 electrically silent AH-cells that were injected with neurobiotin were found to be multipolar Dogiel type II neurons. In contrast, when recordings were made from myenteric S-neurons, two distinct electrical patterns of electrical activity were recorded. Recordings from 25 of 48 S-neurons showed spontaneous FEPSPs, the majority of which (22 of 25) showed periods when discrete clusters of FEPSPs (mean duration 88 ms) could be temporally correlated with the onset of EJPs or anal IJPs in the CM. Nine S-neurons were electrically quiescent. The second distinct electrical pattern in 14 S-neurons consisted of bursts, or prolonged trains of action potentials, which could be reduced to proximal process potentials (PPPs) in six of these 14 neurons during membrane hyperpolarization. Unlike FEPSPs, PPPs were resistant to a low Ca(2+)-high Mg(2+) solution and did not change in amplitude during hyperpolarizing pulses. Mechanosensory S-neurons were found to be uniaxonal or pseudounipolar filamentous neurons, with morphologies consistent with interneurons. No slow EPSPs were ever recorded from AH- or S-type neurons when IJPs or EJPs occurred in the CM. In summary, we have identified a population of mechanosensory S-neurons in the myenteric plexus of the distal colon which appear to be largely stretch sensitive, rather than muscle-tension sensitive, since they generate ongoing trains of action potentials in the presence of nifedipine. No evidence was found to suggest that in paralysed preparations, the repetitive firing in ascending excitatory or descending inhibitory nerve pathways was initiated by myenteric AH-neurons, or slow synaptic transmission.
Figures


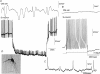



References
-
- Brookes SJ. Classes of enteric nerve cell. Anat Record. 2001;262:58–70. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

