PI3 kinase is important for Ras, MEK and Erk activation of Epo-stimulated human erythroid progenitors
- PMID: 15149544
- PMCID: PMC419721
- DOI: 10.1186/1741-7007-2-7
PI3 kinase is important for Ras, MEK and Erk activation of Epo-stimulated human erythroid progenitors
Abstract
Background: Erythropoietin is a multifunctional cytokine which regulates the number of erythrocytes circulating in mammalian blood. This is crucial in order to maintain an appropriate oxygen supply throughout the body. Stimulation of primary human erythroid progenitors (PEPs) with erythropoietin (Epo) leads to the activation of the mitogenic kinases (MEKs and Erks). How this is accomplished mechanistically remained unclear.
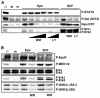
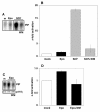
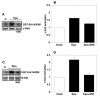
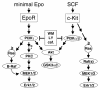
Results: Biochemical studies with human cord blood-derived PEPs now show that Ras and the class Ib enzyme of the phosphatidylinositol-3 kinase (PI3K) family, PI3K gamma, are activated in response to minimal Epo concentrations. Surprisingly, three structurally different PI3K inhibitors block Ras, MEK and Erk activation in PEPs by Epo. Furthermore, Erk activation in PEPs is insensitive to the inhibition of Raf kinases but suppressed upon PKC inhibition. In contrast, Erk activation induced by stem cell factor, which activates c-Kit in the same cells, is sensitive to Raf inhibition and insensitive to PI3K and PKC inhibitors.
Conclusions: These unexpected findings contrast with previous results in human primary cells using Epo at supraphysiological concentrations and open new doors to eventually understanding how low Epo concentrations mediate the moderate proliferation of erythroid progenitors under homeostatic blood oxygen levels. They indicate that the basal activation of MEKs and Erks in PEPs by minimal concentrations of Epo does not occur through the classical cascade Shc/Grb2/Sos/Ras/Raf/MEK/Erk. Instead, MEKs and Erks are signal mediators of PI3K, probably the recently described PI3K gamma, through a Raf-independent signaling pathway which requires PKC activity. It is likely that higher concentrations of Epo that are induced by hypoxia, for example, following blood loss, lead to additional mitogenic signals which greatly accelerate erythroid progenitor proliferation.
Figures



Similar articles
-
Constitutive activation of the MEK/ERK pathway mediates all effects of oncogenic H-ras expression in primary erythroid progenitors.Blood. 2004 Sep 15;104(6):1679-87. doi: 10.1182/blood-2004-04-1362. Epub 2004 May 27. Blood. 2004. PMID: 15166036
-
Activation of phosphatidylinositol 3-kinase is important for erythropoietin-induced erythropoiesis from CD34(+) hematopoietic progenitor cells.Exp Hematol. 2002 Sep;30(9):990-1000. doi: 10.1016/s0301-472x(02)00868-8. Exp Hematol. 2002. PMID: 12225790
-
Critical role for PI 3-kinase in the control of erythropoietin-induced erythroid progenitor proliferation.Blood. 2003 May 1;101(9):3436-43. doi: 10.1182/blood-2002-07-2332. Epub 2002 Dec 27. Blood. 2003. PMID: 12506011
-
Emerging EPO and EPO receptor regulators and signal transducers.Blood. 2015 Jun 4;125(23):3536-41. doi: 10.1182/blood-2014-11-575357. Epub 2015 Apr 17. Blood. 2015. PMID: 25887776 Free PMC article. Review.
-
RAS and downstream RAF-MEK and PI3K-AKT signaling in neuronal development, function and dysfunction.Biol Chem. 2016 Mar;397(3):215-22. doi: 10.1515/hsz-2015-0270. Biol Chem. 2016. PMID: 26760308 Free PMC article. Review.
Cited by
-
Signal strength dictates phosphoinositide 3-kinase contribution to Ras/extracellular signal-regulated kinase 1 and 2 activation via differential Gab1/Shp2 recruitment: consequences for resistance to epidermal growth factor receptor inhibition.Mol Cell Biol. 2008 Jan;28(2):587-600. doi: 10.1128/MCB.01318-07. Epub 2007 Nov 19. Mol Cell Biol. 2008. PMID: 18025104 Free PMC article.
-
RAS Interaction with PI3K: More Than Just Another Effector Pathway.Genes Cancer. 2011 Mar;2(3):261-74. doi: 10.1177/1947601911408079. Genes Cancer. 2011. PMID: 21779497 Free PMC article.
-
AG490 suppresses EPO-mediated activation of JAK2-STAT but enhances blood flow recovery in rats with critical limb ischemia.J Inflamm (Lond). 2016 Jun 4;13:18. doi: 10.1186/s12950-016-0126-3. eCollection 2016. J Inflamm (Lond). 2016. PMID: 27274711 Free PMC article.
-
Microarray and proteomics analyses of human intestinal epithelial cells treated with the Aeromonas hydrophila cytotoxic enterotoxin.Infect Immun. 2005 May;73(5):2628-43. doi: 10.1128/IAI.73.5.2628-2643.2005. Infect Immun. 2005. PMID: 15845465 Free PMC article.
-
Core erythropoietin receptor signals for late erythroblast development.Blood. 2006 Apr 1;107(7):2662-72. doi: 10.1182/blood-2005-02-0684. Epub 2005 Dec 6. Blood. 2006. PMID: 16332976 Free PMC article.
References
-
- Klingmuller U. The role of tyrosine phosphorylation in proliferation and maturation of erythroid progenitor cells--signals emanating from the erythropoietin receptor. Eur J Biochem. 1997;249:637–647. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

