Completion of neuronal migration regulated by loss of Ca(2+) transients
- PMID: 15150416
- PMCID: PMC420419
- DOI: 10.1073/pnas.0401000101
Completion of neuronal migration regulated by loss of Ca(2+) transients
Abstract
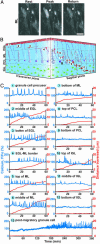
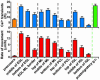
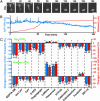
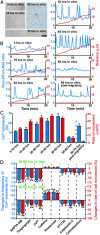
The migration of immature neurons constitutes one of the major processes by which the central nervous system takes shape. Completing the migration at the final destination requires the loss of cell body motility, but little is known about the signaling mechanisms underlying this process. Here, we show that a loss of transient Ca(2+) elevations triggers the completion of cerebellar granule cell migration. Simultaneous observation of the intracellular Ca(2+) levels and cell movement in cerebellar slices of the early postnatal mice revealed that granule cells exhibit distinct frequencies of the transient Ca(2+) elevations as they migrate in different cortical layers, and complete the migration only after the loss of Ca(2+) elevations. The reduction of the Ca(2+) elevation frequency by decreasing Ca(2+) influx, or by inhibiting the activity of phospholipase C, PKC, or Ca(2+)/calmodulin, halted the granule cell movement prematurely. In contrast, increasing the Ca(2+) elevation frequency by increasing Ca(2+) release from internal stores, or by elevating intracellular cAMP levels, significantly delayed the completion of granule cell migration. The timing of the loss of Ca(2+) elevations was intrinsically set in the granule cells and influenced by external cues. These results suggest that Ca(2+) signaling, dictated by multiple signaling systems, functions as a mediator for completing the migration of immature neurons.
Figures
References
-
- Rakic, P. (1990) Experientia 46, 882-891. - PubMed
-
- Hatten, M. E. (1999) Annu. Rev. Neurosci. 22, 511-539. - PubMed
-
- Caviness, V. S., Jr., & Rakic, P. (1978) Annu. Rev. Neurosci. 1, 297-326. - PubMed
-
- Ross, M. E. & Walsh, C. A. (2001) Annu. Rev. Neurosci. 24, 1041-1070. - PubMed
-
- Rakic, P. (1971) J. Comp. Neurol. 141, 283-312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

