In vivo evaluation of [18F]fluoroetanidazole as a new marker for imaging tumour hypoxia with positron emission tomography
- PMID: 15150578
- PMCID: PMC2409496
- DOI: 10.1038/sj.bjc.6601862
In vivo evaluation of [18F]fluoroetanidazole as a new marker for imaging tumour hypoxia with positron emission tomography
Abstract
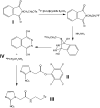
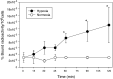
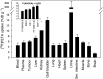
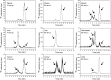
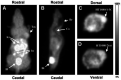
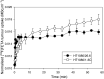
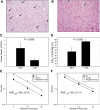
Development of hypoxia-targeted therapies has stimulated the search for clinically applicable noninvasive markers of tumour hypoxia. Here, we describe the validation of [(18)F]fluoroetanidazole ([(18)F]FETA) as a tumour hypoxia marker by positron emission tomography (PET). Cellular transport and retention of [(18)F]FETA were determined in vitro under air vs nitrogen. Biodistribution and metabolism of the radiotracer were determined in mice bearing MCF-7, RIF-1, EMT6, HT1080/26.6, and HT1080/1-3C xenografts. Dynamic PET imaging was performed on a dedicated small animal scanner. [(18)F]FETA, with an octanol-water partition coefficient of 0.16+/-0.01, was selectively retained by RIF-1 cells under hypoxia compared to air (3.4- to 4.3-fold at 60-120 min). The radiotracer was stable in the plasma and distributed well to all the tissues studied. The 60-min tumour/muscle ratios positively correlated with the percentage of pO(2) values <5 mmHg (r=0.805, P=0.027) and carbogen breathing decreased [(18)F]FETA-derived radioactivity levels (P=0.028). In contrast, nitroreductase activity did not influence accumulation. Tumours were sufficiently visualised by PET imaging within 30-60 min. Higher fractional retention of [(18)F]FETA in HT1080/1-3C vs HT1080/26.6 tumours determined by dynamic PET imaging (P=0.05) reflected higher percentage of pO(2) values <1 mmHg (P=0.023), lower vessel density (P=0.026), and higher radiobiological hypoxic fraction (P=0.008) of the HT1080/1-3C tumours. In conclusion, [(18)F]FETA shows hypoxia-dependent tumour retention and is, thus, a promising PET marker that warrants clinical evaluation.
Figures

Similar articles
-
Characterization of [18F]fluoroetanidazole, a new radiopharmaceutical for detecting tumor hypoxia.J Nucl Med. 1999 Jun;40(6):1072-9. J Nucl Med. 1999. PMID: 10452326
-
Comparison of three 18F-labeled 2-nitroimidazoles for imaging hypoxia in breast cancer xenografts: [18F]FBNA, [18F]FAZA and [18F]FMISO.Nucl Med Biol. 2023 Sep-Oct;124-125:108383. doi: 10.1016/j.nucmedbio.2023.108383. Epub 2023 Aug 26. Nucl Med Biol. 2023. PMID: 37651917
-
3'-deoxy-3'-[18F]fluorothymidine as a new marker for monitoring tumor response to antiproliferative therapy in vivo with positron emission tomography.Cancer Res. 2003 Jul 1;63(13):3791-8. Cancer Res. 2003. PMID: 12839975
-
[18F]Fluoroetanidazole.2005 Sep 28 [updated 2005 Oct 19]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2005 Sep 28 [updated 2005 Oct 19]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 20641756 Free Books & Documents. Review.
-
Non-invasive PET and SPECT imaging of tissue hypoxia using isotopically labeled 2-nitroimidazoles.Adv Exp Med Biol. 2003;510:285-92. doi: 10.1007/978-1-4615-0205-0_47. Adv Exp Med Biol. 2003. PMID: 12580442 Review.
Cited by
-
Multimodality imaging of hypoxia in preclinical settings.Q J Nucl Med Mol Imaging. 2010 Jun;54(3):259-80. Q J Nucl Med Mol Imaging. 2010. PMID: 20639813 Free PMC article. Review.
-
Molecular mechanisms of hypoxia in cancer.Clin Transl Imaging. 2017;5(3):225-253. doi: 10.1007/s40336-017-0231-1. Epub 2017 May 11. Clin Transl Imaging. 2017. PMID: 28596947 Free PMC article. Review.
-
Contribution of hypoxia-measuring molecular imaging techniques to radiotherapy planning and treatment.Clin Transl Oncol. 2010 Jan;12(1):22-6. doi: 10.1007/s12094-010-0462-3. Clin Transl Oncol. 2010. PMID: 20080467 Review.
-
PET radiopharmaceuticals for imaging of tumor hypoxia: a review of the evidence.Am J Nucl Med Mol Imaging. 2014 Jun 7;4(4):365-84. eCollection 2014. Am J Nucl Med Mol Imaging. 2014. PMID: 24982822 Free PMC article. Review.
-
The radiation response of cells from 9L gliosarcoma tumours is correlated with [F18]-EF5 uptake.Int J Radiat Biol. 2009 Dec;85(12):1137-47. doi: 10.3109/09553000903242172. Int J Radiat Biol. 2009. PMID: 19995239 Free PMC article.
References
-
- Aboagye EO, Kelson AB, Tracy M, Workman P (1998) Preclinical development and current status of the fluorinated 2-nitroimidazole hypoxia prob N-(2-hydroxy-3,3,3-trifluoropropyl)-2-(2-nitro-1-imidazolyl) acetamide (SR 4554, CRC 94/17): a non-invasive diagnostic probe for the measurement of tumor hypoxia by magnetic resonance spectroscopy and imaging, and by positron emission tomography. Anti-Cancer Drug Des 13: 703–730 - PubMed
-
- Aboagye EO, Lewis AD, Tracy M, Workman P (1997a) Bioreductive metabolism of the novel fluorinated 2-nitroimidazole hypoxia probe N-(2-hydroxy-3,3,3-trifluoropropyl)-2-(2-nitroimidazolyl) acetamide (SR-4554). Biochem Pharmacol 54: 1217–1224 - PubMed
-
- Aboagye EO, Maxwell RJ, Kelson AB, Tracy M, Lewis AD, Graham MA, Horsman MR, Griffiths JR, Workman P (1997b) Preclinical evaluation of the fluorinated 2-nitroimidazole N-(2-hydroxy-3,3,3-trifluoropropyl)-2-(2-nitro-1-imidazolyl) acetamide (SR-4554) as a probe for the measurement of tumor hypoxia. Cancer Res 57: 3314–3318 - PubMed
-
- Adams GE, Clarke ED, Flockhart IR, Jacobs RS, Sehmi DS, Stratford IJ, Wardman P, Watts ME, Parrick J, Wallace RG, Smithen CE (1979) Structure–activity relationship in the development of hypoxic cell radiosensitizers. I. Sensitization efficiency. Int J Radiol Biol 35: 133–150 - PubMed
-
- Aebersold DM, Burri P, Beer KT, Laissue J, Djonov V, Greiner RH, Semenza GL (2001) Expression of hypoxia-inducible factor-1alpha: a novel predictive and prognostic parameter in the radiotherapy of oropharyngeal cancer. Cancer Res 61: 2911–2916 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

