Efficient T-cell surveillance of the CNS requires expression of the CXC chemokine receptor 3
- PMID: 15152045
- PMCID: PMC6729455
- DOI: 10.1523/JNEUROSCI.0123-04.2004
Efficient T-cell surveillance of the CNS requires expression of the CXC chemokine receptor 3
Abstract
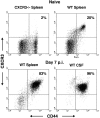
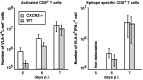
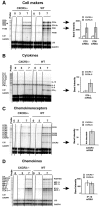
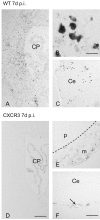
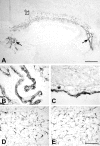
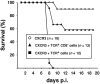
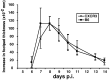
T-cells play an important role in controlling viral infections inside the CNS. To study the role of the chemokine receptor CXCR3 in the migration and positioning of virus-specific effector T-cells within the brain, CXCR3-deficient mice were infected intracerebrally with lymphocytic choriomeningitis virus (LCMV). Analysis of the induction phase of the antiviral CD8+ T-cell response did not reveal any immune defects in CXCR3-deficient mice. Yet, when mice were challenged with LCMV intracerebrally, most CXCR3-deficient mice survived the infection, whereas wild-type mice invariably died from CD8+ T-cell-mediated immunopathology. Quantitative analysis of the cellular infiltrate in CSF of infected mice revealed modest, if any, decrease in the number of mononuclear cells recruited to the meninges in the absence of CXCR3. However, immunohistological analysis disclosed a striking impairment of CD8+ T-cells from CXCR3-deficient mice to migrate from the meninges into the outer layers of the brain parenchyma despite similar localization of virus-infected target cells. Reconstitution of CXCR3-deficient mice with wild-type CD8+ T-cells completely restored susceptibility to LCMV-induced meningitis. Thus, taken together, our results strongly point to a critical role for CXCR3 in the positioning of effector T-cells at sites of viral inflammation in the brain.
Figures

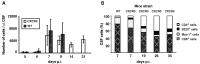
References
-
- Andersson EC, Christensen JP, Marker O, Thomsen AR (1994) Changes in cell adhesion molecule expression on T cells associated with systemic virus infection. J Immunol 152: 1237–1245. - PubMed
-
- Andreasen SO, Christensen JP, Marker O, Thomsen AR (1999) Virus-induced non-specific signals cause cell cycle progression of primed CD8(+) T cells but do not induce cell differentiation. Int Immunol 11: 1463–1473. - PubMed
-
- Asensio VC, Kincaid C, Campbell IL (1999) Chemokines and the inflammatory response to viral infection in the central nervous system with a focus on lymphocytic choriomeningitis virus. J Neurovirol 5: 65–75. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
