The Arg451Cys-neuroligin-3 mutation associated with autism reveals a defect in protein processing
- PMID: 15152050
- PMCID: PMC6729460
- DOI: 10.1523/JNEUROSCI.0468-04.2004
The Arg451Cys-neuroligin-3 mutation associated with autism reveals a defect in protein processing
Abstract
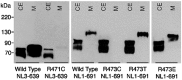
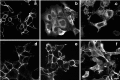
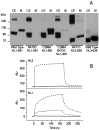
The neuroligins are a family of postsynaptic transmembrane proteins that associate with presynaptic partners, the beta-neurexins. Neurexins and neuroligins play a critical role in initiating formation and differentiation of synaptic junctions. A recent study reported that a mutation of neuroligin-3 (NL3), an X-linked gene, was found in siblings with autistic spectrum disorder in which two affected brothers had a point mutation that substituted a Cys for Arg451. To characterize the mutation at the biochemical level, we analyzed expression and activity of the mutated protein. Mass spectrometry comparison of the disulfide bonding pattern between the native and the mutated proteins indicates the absence of aberrant disulfide bonding, suggesting that the secondary structure of the mutated protein is conserved. However, the mutation separately affects protein expression and activity. The Cys mutation causes defective neuroligin trafficking, leading to retention of the protein in the endoplasmic reticulum. This, in turn, decreases the delivery of NL3 to the cell surface. Also, the small fraction of protein that reaches the cell membrane lacks or has markedly diminished beta-neurexin-1 (NX1beta) binding activity. Other substitutions for Arg451 allow for normal cellular expression but diminished affinity for NX1beta. Our findings reveal a cellular phenotype and loss of function for a congenital mutation associated with autistic spectrum disorders.
Figures
Similar articles
-
Characterization of the interaction of a recombinant soluble neuroligin-1 with neurexin-1beta.J Biol Chem. 2003 Dec 12;278(50):50497-505. doi: 10.1074/jbc.M306803200. Epub 2003 Sep 30. J Biol Chem. 2003. PMID: 14522992
-
Dissection of synapse induction by neuroligins: effect of a neuroligin mutation associated with autism.J Biol Chem. 2005 Jun 10;280(23):22365-74. doi: 10.1074/jbc.M410723200. Epub 2005 Mar 29. J Biol Chem. 2005. PMID: 15797875
-
Trafficking of cholinesterases and neuroligins mutant proteins. An association with autism.Chem Biol Interact. 2008 Sep 25;175(1-3):349-51. doi: 10.1016/j.cbi.2008.04.023. Epub 2008 Apr 29. Chem Biol Interact. 2008. PMID: 18555979 Free PMC article.
-
Neurexins.Genome Biol. 2013;14(9):213. doi: 10.1186/gb-2013-14-9-213. Genome Biol. 2013. PMID: 24083347 Free PMC article. Review.
-
A review on the current neuroligin mouse models.Sheng Li Xue Bao. 2012 Oct 25;64(5):550-62. Sheng Li Xue Bao. 2012. PMID: 23090496 Review.
Cited by
-
A genetic study of autism in Costa Rica: multiple variables affecting IQ scores observed in a preliminary sample of autistic cases.BMC Psychiatry. 2005 Mar 21;5:15. doi: 10.1186/1471-244X-5-15. BMC Psychiatry. 2005. PMID: 15780135 Free PMC article.
-
Genetic aspects of autism spectrum disorders: insights from animal models.Front Cell Neurosci. 2014 Feb 24;8:58. doi: 10.3389/fncel.2014.00058. eCollection 2014. Front Cell Neurosci. 2014. PMID: 24605088 Free PMC article. Review.
-
Gene Disrupting Mutations Associated with Regression in Autism Spectrum Disorder.J Autism Dev Disord. 2017 Nov;47(11):3600-3607. doi: 10.1007/s10803-017-3256-4. J Autism Dev Disord. 2017. PMID: 28856484 Free PMC article.
-
A neuroligin-3 mutation implicated in autism increases inhibitory synaptic transmission in mice.Science. 2007 Oct 5;318(5847):71-6. doi: 10.1126/science.1146221. Epub 2007 Sep 6. Science. 2007. PMID: 17823315 Free PMC article.
-
Autism-associated R451C mutation in neuroligin3 leads to activation of the unfolded protein response in a PC12 Tet-On inducible system.Biochem J. 2016 Feb 15;473(4):423-34. doi: 10.1042/BJ20150274. Epub 2015 Nov 30. Biochem J. 2016. PMID: 26621873 Free PMC article.
References
-
- Comoletti D, Flynn RE, Jennings LL, Chubykin A, Matsumura T, Hasegawa H, Südhof TC, Taylor P (2003) Characterization of the interaction of a recombinant soluble neuroligin-1 with neurexin-1beta. J Biol Chem 278: 50497–50505. - PubMed
-
- Davis AC, Shulman MJ (1989) IgM–molecular requirements for its assembly and function. Immunol Today 10: 118–122. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
