Clostridium sordellii lethal toxin is maintained in a multimeric protein complex
- PMID: 15155642
- PMCID: PMC415707
- DOI: 10.1128/IAI.72.6.3366-3372.2004
Clostridium sordellii lethal toxin is maintained in a multimeric protein complex
Erratum in
- Infect Immun. 2004 Aug;72(8):4932
Abstract
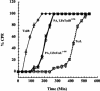
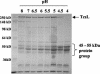
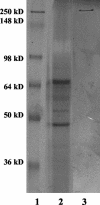
Clostridium sordellii lethal toxin (TcsL) is distinct among large clostridial toxins (LCTs), as it is markedly reduced in its rate of intoxication at pH 8.0 yet is cytotoxic at pH 4.0. Results from the present study suggest that TcsL's slow rate of intoxication at pH 8.0 is linked to formation of a high-molecular-weight complex containing dissociable pH 4.0-sensitive polypeptides. The cytosolic delivery of TcsL's enzymatic domain by using a surrogate cell entry system resulted in cytopathic effect rates similar to those of other LCTs at pH 8.0, further indicating that rate-limiting steps occurred at the point of cell entry. Since these rate-limiting steps could be overcome at pH 4.0, TcsL was examined across a range of pH values and was found to dissociate into distinct 45- to 55-kDa polypeptides between pH 4.0 and pH 5.0. The polypeptides reassociated when shifted back to pH 8.0. At pH 8.0, this complex was resistant to sodium dodecyl sulfate (SDS) and multiple proteases; however, following dissociation, the polypeptides became protease sensitive. Dissociation of TcsL, and cytotoxicity, could be blocked by preincubation with ethylene glycol bis(sulfosuccinimidylsuccinate), resulting in cross-linking of the polypeptides. TcsL was also examined at pH 8.0 by using SDS-agarose gel electrophoresis and transmission electron microscopy and was found to exist in a higher-molecular-weight complex which resolved at a size exceeding 750 kDa and also dissociated at pH 4.0. However, this complex did not reassemble following a shift back to pH 8.0. Collectively, these data suggest that TcsL is maintained in a protease-resistant, high-molecular-weight complex, which dissociates at pH 4.0, leading to cytotoxicity.
Figures

Similar articles
-
pH-enhanced cytopathic effects of Clostridium sordellii lethal toxin.Infect Immun. 2001 Sep;69(9):5487-93. doi: 10.1128/IAI.69.9.5487-5493.2001. Infect Immun. 2001. PMID: 11500421 Free PMC article.
-
The Tip of the Four N-Terminal α-Helices of Clostridium sordellii Lethal Toxin Contains the Interaction Site with Membrane Phosphatidylserine Facilitating Small GTPases Glucosylation.Toxins (Basel). 2016 Mar 25;8(4):90. doi: 10.3390/toxins8040090. Toxins (Basel). 2016. PMID: 27023605 Free PMC article.
-
Killing of rat basophilic leukemia cells by lethal toxin from Clostridium sordellii: critical role of phosphatidylinositide 3'-OH kinase/Akt signaling.Biochemistry. 2009 Mar 3;48(8):1785-92. doi: 10.1021/bi800708b. Biochemistry. 2009. PMID: 19199813
-
Large Clostridial Toxins: Mechanisms and Roles in Disease.Microbiol Mol Biol Rev. 2021 Aug 18;85(3):e0006421. doi: 10.1128/MMBR.00064-21. Epub 2021 Jun 2. Microbiol Mol Biol Rev. 2021. PMID: 34076506 Free PMC article. Review.
-
Functional implications of lethal toxin-catalysed glucosylation of (H/K/N)Ras and Rac1 in Clostridium sordellii-associated disease.Eur J Cell Biol. 2011 Nov;90(11):959-65. doi: 10.1016/j.ejcb.2010.10.009. Epub 2010 Dec 4. Eur J Cell Biol. 2011. PMID: 21134703 Review.
Cited by
-
The Sialidase NanS Enhances Non-TcsL Mediated Cytotoxicity of Clostridium sordellii.Toxins (Basel). 2016 Jun 17;8(6):189. doi: 10.3390/toxins8060189. Toxins (Basel). 2016. PMID: 27322322 Free PMC article.
-
The alternative translational profile that underlies the immune-evasive state of persistence in Chlamydiaceae exploits differential tryptophan contents of the protein repertoire.Microbiol Mol Biol Rev. 2012 Jun;76(2):405-43. doi: 10.1128/MMBR.05013-11. Microbiol Mol Biol Rev. 2012. PMID: 22688818 Free PMC article. Review.
-
Chlamydia exploit the mammalian tryptophan-depletion defense strategy as a counter-defensive cue to trigger a survival state of persistence.Front Cell Infect Microbiol. 2014 Feb 28;4:17. doi: 10.3389/fcimb.2014.00017. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 24616884 Free PMC article. Review.
-
Plasma membrane association of three classes of bacterial toxins is mediated by a basic-hydrophobic motif.Cell Microbiol. 2012 Feb;14(2):286-98. doi: 10.1111/j.1462-5822.2011.01718.x. Epub 2011 Nov 29. Cell Microbiol. 2012. PMID: 22044757 Free PMC article.
-
Inositol hexakisphosphate-dependent processing of Clostridium sordellii lethal toxin and Clostridium novyi alpha-toxin.J Biol Chem. 2011 Apr 29;286(17):14779-86. doi: 10.1074/jbc.M110.200691. Epub 2011 Mar 8. J Biol Chem. 2011. PMID: 21385871 Free PMC article.
References
-
- Adamkiewicz, T. V., D. Goodman, B. Burke, D. M. Lyerly, J. Goswitz, and P. Ferrieri. 1993. Neonatal Clostridium sordellii toxic omphalitis. Pediatr. Infect. Dis. J. 12:253-257. - PubMed
-
- Ahmed, S. A., P. McPhie, and L. A. Smith. 2003. Autocatalytically fragmented light chain of botulinum A neurotoxin is enzymatically active. Biochemistry 42:12539-12549. - PubMed
-
- Aktories, K., G. Schmidt, and I. Just. 2000. Rho GTPases as targets of bacterial protein toxins. Biol. Chem. 381:421-426. - PubMed
-
- Amimoto, K., E. Oishi, H. Yasuhar, O. Sasak, S. Katayama, T. Kitajima, A. Izumida, and T. Hirahara. 2001. Protective effects of Clostridium sordellii LT and HT toxoids against challenge with spores in guinea pigs. J. Vet. Med. Sci. 63:879-883. - PubMed
-
- Boquet, P. 1999. Bacterial toxins inhibiting or activating small GTP-binding proteins. Ann. N. Y. Acad. Sci. 886:83-90. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

