Interaction of Blastomyces dermatitidis, Sporothrix schenckii, and Histoplasma capsulatum with Acanthamoeba castellanii
- PMID: 15155655
- PMCID: PMC415723
- DOI: 10.1128/IAI.72.6.3478-3488.2004
Interaction of Blastomyces dermatitidis, Sporothrix schenckii, and Histoplasma capsulatum with Acanthamoeba castellanii
Abstract
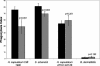
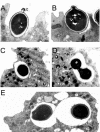
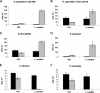
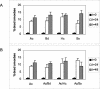
Several dimorphic fungi are important human pathogens, but the origin and maintenance of virulence in these organisms is enigmatic, since an interaction with a mammalian host is not a requisite for fungal survival. Recently, Cryptococcus neoformans was shown to interact with macrophages, slime molds, and amoebae in a similar manner, suggesting that fungal pathogenic strategies may arise from environmental interactions with phagocytic microorganisms. In this study, we examined the interactions of three dimorphic fungi with the soil amoeba Acanthameobae castellanii. Yeast forms of Blastomyces dermatitidis, Sporothrix schenckii, and Histoplasma capsulatum were each ingested by amoebae and macrophages, and phagocytosis of yeast cells resulted in amoeba death and fungal growth. H. capsulatum conidia were also cytotoxic to amoebae. For each fungal species, exposure of yeast cells to amoebae resulted in an increase in hyphal cells. Exposure of an avirulent laboratory strain of H. capsulatum to A. castellanii selected for, or induced, a phenotype of H. capsulatum that caused a persistent murine lung infection. These results are consistent with the view that soil amoebae may contribute to the selection and maintenance of certain traits in pathogenic dimorphic fungi that confer on these microbes the capacity for virulence in mammals.
Figures




Similar articles
-
Interaction of Talaromyces marneffei with free living soil amoeba as a model of fungal pathogenesis.Front Cell Infect Microbiol. 2022 Oct 3;12:1023067. doi: 10.3389/fcimb.2022.1023067. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36262181 Free PMC article.
-
Unravelling the interactions of the environmental host Acanthamoeba castellanii with fungi through the recognition by mannose-binding proteins.Cell Microbiol. 2019 Oct;21(10):e13066. doi: 10.1111/cmi.13066. Epub 2019 Jun 19. Cell Microbiol. 2019. PMID: 31173452
-
Unraveling the secrets of Histoplasma capsulatum. A model to study morphogenic adaptation during parasite host/host interaction.Verh K Acad Geneeskd Belg. 1995;57(2):133-56. Verh K Acad Geneeskd Belg. 1995. PMID: 7571855 Review.
-
Rapid conversion of Histoplasma capsulatum, Blastomyces dermatitidis and sporothrix schenckii in tissue culture.J Clin Microbiol. 1975 May;1(5):420-4. doi: 10.1128/jcm.1.5.420-424.1975. J Clin Microbiol. 1975. PMID: 1236872 Free PMC article.
-
[Dimorphism and pathogenesis of Histoplasma capsulatum].Rev Argent Microbiol. 2006 Oct-Dec;38(4):235-42. Rev Argent Microbiol. 2006. PMID: 17370580 Review. Spanish.
Cited by
-
Mimivirus Fibrils Are Important for Viral Attachment to the Microbial World by a Diverse Glycoside Interaction Repertoire.J Virol. 2015 Dec;89(23):11812-9. doi: 10.1128/JVI.01976-15. Epub 2015 Sep 16. J Virol. 2015. PMID: 26378162 Free PMC article.
-
Phagocytosis of Cryptococcus neoformans by, and nonlytic exocytosis from, Acanthamoeba castellanii.Appl Environ Microbiol. 2010 Sep;76(18):6056-62. doi: 10.1128/AEM.00812-10. Epub 2010 Jul 30. Appl Environ Microbiol. 2010. PMID: 20675457 Free PMC article.
-
Concurrent amoebic and histoplasma colitis: a rare cause of massive lower gastrointestinal bleeding.World J Gastroenterol. 2010 Mar 14;16(10):1296-8. doi: 10.3748/wjg.v16.i10.1296. World J Gastroenterol. 2010. PMID: 20222177 Free PMC article.
-
A hidden battle in the dirt: Soil amoebae interactions with Paracoccidioides spp.PLoS Negl Trop Dis. 2019 Oct 7;13(10):e0007742. doi: 10.1371/journal.pntd.0007742. eCollection 2019 Oct. PLoS Negl Trop Dis. 2019. PMID: 31589617 Free PMC article.
-
Dictyostelium discoideum as a Novel Host System to Study the Interaction between Phagocytes and Yeasts.Front Microbiol. 2016 Oct 21;7:1665. doi: 10.3389/fmicb.2016.01665. eCollection 2016. Front Microbiol. 2016. PMID: 27818653 Free PMC article.
References
-
- Atkinson, J. B., and T. L. McCurley. 1983. Pulmonary blastomycosis: filamentous forms in an immunocompromised patient with fulminating respiratory failure. Hum. Pathol. 14:186-188. - PubMed
-
- Brass, C., C. M. Volkmann, D. E. Philpott, H. P. Klein, C. J. Halde, and D. A. Stevens. 1982. Spontaneous mutant of Blastomyces dermatitidis attenuated in virulence for mice. Sabouraudia 20:145-158. - PubMed
-
- Brummer, E., A. Restrepo, L. H. Hanson, and D. A. Stevens. 1990. Virulence of Paracoccidiodes brasiliensis: the influence of in vitro passage and storage. Mycopathologia 109:13-17. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

