Ferritin reactions: direct identification of the site for the diferric peroxide reaction intermediate
- PMID: 15166287
- PMCID: PMC423233
- DOI: 10.1073/pnas.0401146101
Ferritin reactions: direct identification of the site for the diferric peroxide reaction intermediate
Abstract
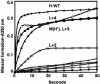
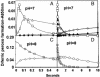
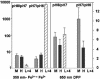
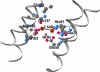
Ferritins managing iron-oxygen biochemistry in animals, plants, and microorganisms belong to the diiron carboxylate protein family and concentrate iron as ferric oxide approximately 10(14) times above the ferric K(s). Ferritin iron (up to 4,500 atoms), used for iron cofactors and heme, or to trap DNA-damaging oxidants in microorganisms, is concentrated in the protein nanocage cavity (5-8 nm) formed during assembly of polypeptide subunits, 24 in maxiferritins and 12 in miniferritins/DNA protection during starvation proteins. Direct identification of ferritin ferroxidase (F(ox)) sites, complicated by multiple types of iron-ferritin interactions, is now achieved with chimeric proteins where putative F(ox) site residues were introduced singly and cumulatively into an inactive host, an L maxiferritin. A dimagnesium ferritin cocrystal model guided site design and the diferric peroxo F(ox) intermediates (A at 650 nm) monitored activity. Diferric peroxo formation in chimeric and WT proteins had similar K(app) values and Hill coefficients. Catalytic activity required cooperative ferrous substrate binding to two sites A (E, EXXH) and B (E, QXXD). The weaker B sites in ferritin contrast with stronger B sites (E, EXXH) in diiron carboxylate oxygenases, explaining diferric oxo/hydroxo product release in ferritin vs. diiron cofactor retention in oxygenases. Codons for Q/H and D/E differ by single nucleotides, suggesting simple DNA mutations relate site B diiron substrate sites and diiron cofactor sites in proteins. The smaller k(cat) values in chimeras indicate the absence of second-shell residues important for ferritin substrate-product channeling that, when identified, will outline the entire iron path from ferritin pores through the F(ox) site to the mineral cavity.
Figures
Similar articles
-
The ferritin Fe2 site at the diiron catalytic center controls the reaction with O2 in the rapid mineralization pathway.Proc Natl Acad Sci U S A. 2008 Nov 25;105(47):18182-7. doi: 10.1073/pnas.0805083105. Epub 2008 Nov 14. Proc Natl Acad Sci U S A. 2008. PMID: 19011101 Free PMC article.
-
The ferroxidase reaction of ferritin reveals a diferric mu-1,2 bridging peroxide intermediate in common with other O2-activating non-heme diiron proteins.Biochemistry. 1999 Apr 27;38(17):5290-5. doi: 10.1021/bi990095l. Biochemistry. 1999. PMID: 10220314
-
Stoichiometric production of hydrogen peroxide and parallel formation of ferric multimers through decay of the diferric-peroxo complex, the first detectable intermediate in ferritin mineralization.Biochemistry. 2002 Nov 12;41(45):13435-43. doi: 10.1021/bi026478s. Biochemistry. 2002. PMID: 12416989
-
The iron redox and hydrolysis chemistry of the ferritins.Biochim Biophys Acta. 2010 Aug;1800(8):719-31. doi: 10.1016/j.bbagen.2010.03.021. Epub 2010 Apr 9. Biochim Biophys Acta. 2010. PMID: 20382203 Review.
-
Mineralization in ferritin: an efficient means of iron storage.J Struct Biol. 1999 Jun 30;126(3):182-94. doi: 10.1006/jsbi.1999.4118. J Struct Biol. 1999. PMID: 10441528 Review.
Cited by
-
Iron homeostasis and post-hemorrhagic hydrocephalus: a review.Front Neurol. 2024 Jan 12;14:1287559. doi: 10.3389/fneur.2023.1287559. eCollection 2023. Front Neurol. 2024. PMID: 38283681 Free PMC article. Review.
-
Computational modeling of the dizinc-ferroxidase complex of human H ferritin: direct comparison of the density functional theory calculated and experimental structures.J Biol Inorg Chem. 2009 Nov;14(8):1199-208. doi: 10.1007/s00775-009-0563-z. Epub 2009 Jul 8. J Biol Inorg Chem. 2009. PMID: 19585161
-
Human mitochondrial ferritin exhibits highly unusual iron-O2 chemistry distinct from that of cytosolic ferritins.Nat Commun. 2025 May 20;16(1):4695. doi: 10.1038/s41467-025-59463-1. Nat Commun. 2025. PMID: 40393986 Free PMC article.
-
Mathematical modeling of the dynamic storage of iron in ferritin.BMC Syst Biol. 2010 Nov 3;4:147. doi: 10.1186/1752-0509-4-147. BMC Syst Biol. 2010. PMID: 21047430 Free PMC article.
-
Role of H-1 and H-2 subunits of soybean seed ferritin in oxidative deposition of iron in protein.J Biol Chem. 2010 Oct 15;285(42):32075-86. doi: 10.1074/jbc.M110.130435. Epub 2010 Aug 11. J Biol Chem. 2010. PMID: 20702403 Free PMC article.
References
-
- Harrison, P. M., Hempstead, P. D., Artymiuk, P. J. & Andrews, S. C. (1998) Met. Ions Biol. Syst. 35, 435–477. - PubMed
-
- Chasteen, N. D. & Harrison, P. M. (1999) J. Struct. Biol. 126, 182–194. - PubMed
-
- Theil, E. C. (2001) in Handbook of Metalloproteins, eds. Messerschmidt, A., Huber, R., Poulos, T. & Wieghardt, K. (Wiley, Chichester, U.K.), pp. 771–781.
-
- Ferreira, F., Bucchini, D., Martin, M. E., Levi, S., Arosio, P., Grandchamp, B. & Beaumont, C. (2000) J. Biol. Chem. 275, 3021–3024. - PubMed
-
- Curtis, A. R., Fey, C., Morris, C. M., Bindoff, L. A., Ince, P. G., Chinnery, P. F., Coulthard, A., Jackson, M. J., Jackson, A. P., McHale, D. P., et al. (2001) Nat. Genet. 28, 350–354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

