A cholinergic synaptically triggered event participates in the generation of persistent activity necessary for eye fixation
- PMID: 15175380
- PMCID: PMC6729203
- DOI: 10.1523/JNEUROSCI.0235-04.2004
A cholinergic synaptically triggered event participates in the generation of persistent activity necessary for eye fixation
Abstract
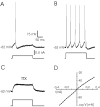
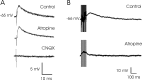
An exciting topic regarding integrative properties of the nervous system is how transient motor commands or brief sensory stimuli are able to evoke persistent neuronal changes, mainly as a sustained, tonic action potential firing. A persisting firing seems to be necessary for postural maintenance after a previous movement. We have studied in vitro and in vivo the generation of the persistent neuronal activity responsible for eye fixation after spontaneous eye movements. Rat sagittal brainstem slices were used for the intracellular recording of prepositus hypoglossi (PH) neurons and their synaptic activation from nearby paramedian pontine reticular formation (PPRF) neurons. Single electrical pulses applied to the PPRF showed a monosynaptic glutamatergic projection on PH neurons, acting on AMPA-kainate receptors. Train stimulation of the PPRF area evoked a sustained depolarization of PH neurons exceeding (by hundreds of milliseconds) stimulus duration. Both duration and amplitude of this sustained depolarization were linearly related to train frequency. The train-evoked sustained depolarization was the result of interaction between glutamatergic excitatory burst neurons and cholinergic mesopontine reticular fibers projecting onto PH neurons, because it was prevented by slice superfusion with cholinergic antagonists and mimicked by cholinergic agonists. As expected, microinjections of cholinergic antagonists in the PH nucleus of alert behaving cats evoked a gaze-holding deficit consisting of a re-centering drift of the eye after each saccade. These findings suggest that a slow, cholinergic, synaptically triggered event participates in the generation of persistent activity characteristic of PH neurons carrying eye position signals.
Figures







Similar articles
-
A cholinergic mechanism underlies persistent neural activity necessary for eye fixation.Prog Brain Res. 2006;154:211-24. doi: 10.1016/S0079-6123(06)54011-7. Prog Brain Res. 2006. PMID: 17010712 Review.
-
Cooperative glutamatergic and cholinergic mechanisms generate short-term modifications of synaptic effectiveness in prepositus hypoglossi neurons.J Neurosci. 2005 Oct 26;25(43):9902-6. doi: 10.1523/JNEUROSCI.2061-05.2005. J Neurosci. 2005. PMID: 16251437 Free PMC article.
-
A cholinergic mechanism for eye fixation.J Mol Neurosci. 2006;30(1-2):125-8. doi: 10.1385/jmn:30:1:125. J Mol Neurosci. 2006. PMID: 17192657
-
Effects of reversible inactivation of the primate mesencephalic reticular formation. I. Hypermetric goal-directed saccades.J Neurophysiol. 2000 Apr;83(4):2260-84. doi: 10.1152/jn.2000.83.4.2260. J Neurophysiol. 2000. PMID: 10758133
-
Studies of the role of the paramedian pontine reticular formation in the control of head-restrained and head-unrestrained gaze shifts.Ann N Y Acad Sci. 2002 Apr;956:85-98. doi: 10.1111/j.1749-6632.2002.tb02811.x. Ann N Y Acad Sci. 2002. PMID: 11960796 Review.
Cited by
-
Mechanics of mouse ocular motor plant quantified by optogenetic techniques.J Neurophysiol. 2015 Sep;114(3):1455-67. doi: 10.1152/jn.00328.2015. Epub 2015 Jun 24. J Neurophysiol. 2015. PMID: 26108953 Free PMC article.
-
Neurons of the dentate molecular layer in the rabbit hippocampus.PLoS One. 2012;7(11):e48470. doi: 10.1371/journal.pone.0048470. Epub 2012 Nov 7. PLoS One. 2012. PMID: 23144890 Free PMC article.
-
Nicotinic acetylcholine receptor-mediated responses in medial vestibular and prepositus hypoglossi nuclei neurons showing distinct neurotransmitter phenotypes.J Neurophysiol. 2016 Jun 1;115(5):2649-57. doi: 10.1152/jn.00852.2015. Epub 2016 Mar 2. J Neurophysiol. 2016. PMID: 26936981 Free PMC article.
-
Intrinsic physiology of identified neurons in the prepositus hypoglossi and medial vestibular nuclei.J Vestib Res. 2011;21(1):33-47. doi: 10.3233/VES-2011-0394. J Vestib Res. 2011. PMID: 21422541 Free PMC article.
-
A Novel Subpopulation of Prepositus Hypoglossi Nucleus Neurons Projecting to the Cerebellar Anterior Vermis and Hemisphere in Rats.eNeuro. 2025 Jul 21;12(7):ENEURO.0130-25.2025. doi: 10.1523/ENEURO.0130-25.2025. Print 2025 Jul. eNeuro. 2025. PMID: 40664511 Free PMC article.
References
-
- Aksay E, Baker R, Seung HS, Tank DW (2000) Anatomy and discharge properties of premotor neurons in the goldfish medulla that have eye-position signals during fixations. J Neurophysiol 84: 1035–1049. - PubMed
-
- Aksay E, Gamkrelidze G, Seung HS, Baker R, Tank DW (2001) In vivo intracellular recording and perturbation of persistent activity in a neuronal integrator. Nat Neurosci 4: 184–193. - PubMed
-
- Arnold DB, Robinson DA, Leigh RJ (1999) Nystagmus induced by pharmacological inactivation of the brainstem ocular motor integrator in monkey. Vision Res 39: 4286–4295. - PubMed
-
- Barmack NH, Baughman RW, Eckenstein FP, Shojaku H (1992) Secondary vestibular cholinergic projection to the cerebellum of rabbit and rat as revealed by choline acetyltransferase immunohistochemistry, retrograde and orthograde tracers. J Comp Neurol 317: 250–270. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
