Chemical genetic screening identifies sulfonamides that raise organellar pH and interfere with membrane traffic
- PMID: 15180825
- PMCID: PMC2494881
- DOI: 10.1111/j.1398-9219.2004.00193.x
Chemical genetic screening identifies sulfonamides that raise organellar pH and interfere with membrane traffic
Abstract
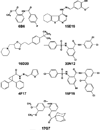
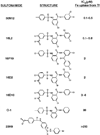
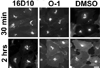
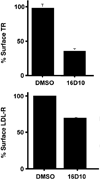
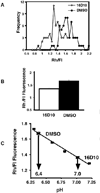
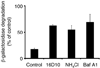
Chemical genetics seeks to identify small molecules that afford functional dissection of cell biological pathways. Previous screens for small molecule inhibitors of exocytic membrane traffic yielded the identification and characterization of several compounds that block traffic from the Golgi to the cell surface as well as transport from the endoplasmic reticulum to the Golgi network [Feng et al. Proc Natl Acad Sci USA 2003;100:6469-6474; Yarrow et al. Comb Chem High Throughput Screen 2003;6:279-286; Feng et al. EMBO Reports 2004: in press]. Here, we screened these inhibitors for potential effects on endocytic membrane traffic. Two structurally related sulfonamides were found to be potent and reversible inhibitors of transferrin-mediated iron uptake. These inhibitors do not block endoplasmic reticulum-to-Golgi transport, but do disrupt Golgi-to-cell surface traffic. The compounds are members of a novel class of sulfonamides that elevate endosomal and lysosomal pH, down-regulate cell surface receptors, and impair recycling of internalized transferrin receptors to the plasma membrane. In vitro experiments revealed that the sulfonamides directly inhibit adenosine triphosphate (ATP) hydrolysis by the V-ATPase and that they also possess a potent proton ionophore activity. While maintenance of organellar pH is known to be a critical factor in both endocytosis and exocytosis, the precise role of acidification, beyond the uncoupling of ligands from their receptors, remains largely unknown. Identification of this novel class of sulfonamide inhibitors provides new chemical tools to better understand the function of organelle pH in membrane traffic and the activity of V-ATPases in particular.
Figures








Similar articles
-
Low cytoplasmic pH inhibits endocytosis and transport from the trans-Golgi network to the cell surface.J Cell Biol. 1989 Feb;108(2):377-87. doi: 10.1083/jcb.108.2.377. J Cell Biol. 1989. PMID: 2918022 Free PMC article.
-
A regulatory role for cAMP-dependent protein kinase in protein traffic along the exocytic route.J Biol Chem. 1996 Nov 29;271(48):30935-41. doi: 10.1074/jbc.271.48.30935. J Biol Chem. 1996. PMID: 8940080
-
Acidification of the endocytic and exocytic pathways.Annu Rev Biochem. 1986;55:663-700. doi: 10.1146/annurev.bi.55.070186.003311. Annu Rev Biochem. 1986. PMID: 2874766 Review. No abstract available.
-
Lysosomes behave as Ca2+-regulated exocytic vesicles in fibroblasts and epithelial cells.J Cell Biol. 1997 Apr 7;137(1):93-104. doi: 10.1083/jcb.137.1.93. J Cell Biol. 1997. PMID: 9105039 Free PMC article.
-
Multiple pathways of exocytosis, endocytosis, and membrane recycling: validation of a Golgi route.Fed Proc. 1983 May 15;42(8):2407-13. Fed Proc. 1983. PMID: 6404654 Review.
Cited by
-
Pharmacology of iron transport.Annu Rev Pharmacol Toxicol. 2013;53:17-36. doi: 10.1146/annurev-pharmtox-010611-134648. Epub 2012 Sep 27. Annu Rev Pharmacol Toxicol. 2013. PMID: 23020294 Free PMC article. Review.
-
Structured cyclic peptides that bind the EH domain of EHD1.Biochemistry. 2014 Jul 29;53(29):4758-60. doi: 10.1021/bi500744q. Epub 2014 Jul 17. Biochemistry. 2014. PMID: 25014215 Free PMC article.
-
The yeast endosomal Na+K+/H+ exchanger Nhx1 regulates cellular pH to control vesicle trafficking.Mol Biol Cell. 2005 Mar;16(3):1396-405. doi: 10.1091/mbc.e04-11-0999. Epub 2005 Jan 5. Mol Biol Cell. 2005. PMID: 15635088 Free PMC article.
-
Identification of novel pathogenic roles of BLZF1/ATF6 in tumorigenesis of gastrointestinal stromal tumor showing Golgi-localized mutant KIT.Cell Death Differ. 2023 Oct;30(10):2309-2321. doi: 10.1038/s41418-023-01220-2. Epub 2023 Sep 13. Cell Death Differ. 2023. PMID: 37704840 Free PMC article.
-
Iron uptake and release by macrophages is sensitive to propranolol.Mol Cell Biochem. 2006 Aug;288(1-2):213-7. doi: 10.1007/s11010-006-9138-2. Epub 2006 May 23. Mol Cell Biochem. 2006. PMID: 16718379
References
-
- Specht KM, Shokat KM. The emerging power of chemical genetics. Curr Opin Cell Biol. 2002;14:155–159. - PubMed
-
- Stockwell BR. Frontiers in chemical genetics. Trends Biotechnol. 2000;18:449–455. - PubMed
-
- Yarrow JC, Feng Y, Perlman ZE, Kirchhausen T, Mitchison TJ. Phenotypic screening of small molecule libraries by high throughput cell imaging. Comb Chem High Throughput Screen. 2003;6:279–286. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 GM036548/GM/NIGMS NIH HHS/United States
- DK57689/DK/NIDDK NIH HHS/United States
- R01 DK057689/DK/NIDDK NIH HHS/United States
- R01 DK064750/DK/NIDDK NIH HHS/United States
- R01 DK052852/DK/NIDDK NIH HHS/United States
- GM36548/GM/NIGMS NIH HHS/United States
- R56 DK052852/DK/NIDDK NIH HHS/United States
- DK33627/DK/NIDDK NIH HHS/United States
- P01 DK055495/DK/NIDDK NIH HHS/United States
- DK52852/DK/NIDDK NIH HHS/United States
- DK56160/DK/NIDDK NIH HHS/United States
- P01 GM062566/GM/NIGMS NIH HHS/United States
- DK55495/DK/NIDDK NIH HHS/United States
- GM62566/GM/NIGMS NIH HHS/United States
- R01 DK056160/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

