Novel role for Netrins in regulating epithelial behavior during lung branching morphogenesis
- PMID: 15186747
- PMCID: PMC2925841
- DOI: 10.1016/j.cub.2004.05.020
Novel role for Netrins in regulating epithelial behavior during lung branching morphogenesis
Abstract
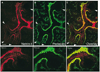
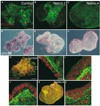
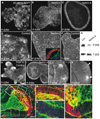
The development of many organs, including the lung, depends upon a process known as branching morphogenesis, in which a simple epithelial bud gives rise to a complex tree-like system of tubes specialized for the transport of gas or fluids. Previous studies on lung development have highlighted a role for fibroblast growth factors (FGFs), made by the mesodermal cells, in promoting the proliferation, budding, and chemotaxis of the epithelial endoderm. Here, by using a three-dimensional culture system, we provide evidence for a novel role for Netrins, best known as axonal guidance molecules, in modulating the morphogenetic response of lung endoderm to exogenous FGFs. This effect involves inhibition of localized changes in cell shape and phosphorylation of the intracellular mitogen-activated protein kinase(s) (ERK1/2, for extracellular signal-regulated kinase-1 and -2), elicited by exogenous FGFs. The temporal and spatial expression of netrin 1, netrin 4, and Unc5b genes and the localization of Netrin-4 protein in vivo suggest a model in which Netrins in the basal lamina locally modulate and fine-tune the outgrowth and shape of emergent epithelial buds.
Conflict of interest statement
M.T.-L. is an inventor on patents or patent applications covering netrin receptors. He is a member of the scientific advisory board and shareholder of Renovis, Inc., and is employed by and is a shareholder in Genentech, Inc. Both companies have a commercial interest in Netrin receptors.
Figures



References
-
- Hogan BL. Morphogenesis. Cell. 1999;96:225–233. - PubMed
-
- Warburton D, Schwarz M, Tefft D, Flores-Delgado G, Anderson KD, Cardoso WV. The molecular basis of lung morphogenesis. Mech. Dev. 2000;92:55–81. - PubMed
-
- Cardoso W. Molecular regulation of lung development. Annu. Rev. Physiol. 2001;63:471–494. - PubMed
-
- Tessier-Lavigne M, Goodman C. The molecular biology of axon guidance. Science. 1996;274:1123–1133. - PubMed
-
- Yu T, Bargmann C. Dynamic regulation of axon guidance. Nat. Neurosci. Suppl. 2001;4:1169–1176. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

